Inflammation as a Source of Disease Biomarkers
Published on: 4 Oct 2021
Inflammation is a broad-ranging biological response that promotes defence, healing and repair in response to injury or illness. It involves a complex network of responses that includes many different biological pathways and cell types throughout the body, particularly within the immune system. As such, it’s also no surprise that many biomarker studies attribute inflammation as a prospective biomarker source (e.g. see recent respiratory disease review by Ibrahim et al.1). But how relevant is inflammation for disease diagnosis, and are inflammation biomarkers really useful?
There are many triggers for inflammation including injury, infection, and irritation. The hallmarks of an inflammatory response include heat, swelling, redness and pain. Typically, inflammation is divided into acute and chronic forms. Acute inflammation normally lasts hours or days and is largely coordinated by neutrophils, while chronic inflammation involves mononucleocytes and can last for years. Long-term chronic inflammation becomes increasingly prevalent with age, can be a factor in cancer progression and can be detrimental to health.
We expect inflammation to produce breath biomarkers
For many inflammatory diseases, current diagnostic methods are not only costly but are also invasive, adding to patient’s stress, and a non-invasive alternative is highly desirable. Inflammation is a component of many of the illnesses which have been studied using breath research (Figure 1). Chronic respiratory and liver diseases, cancers, infections and gastrointestinal illnesses all involve inflammation. Many inflammation processes result in the production and release of small molecules which, either directly or indirectly, can become volatile organic compounds (VOCs) detectable on breath. As such, breath biomarkers could potentially provide cheaper, rapid, and non-invasive screening procedures to diagnose and monitor inflammatory diseases.
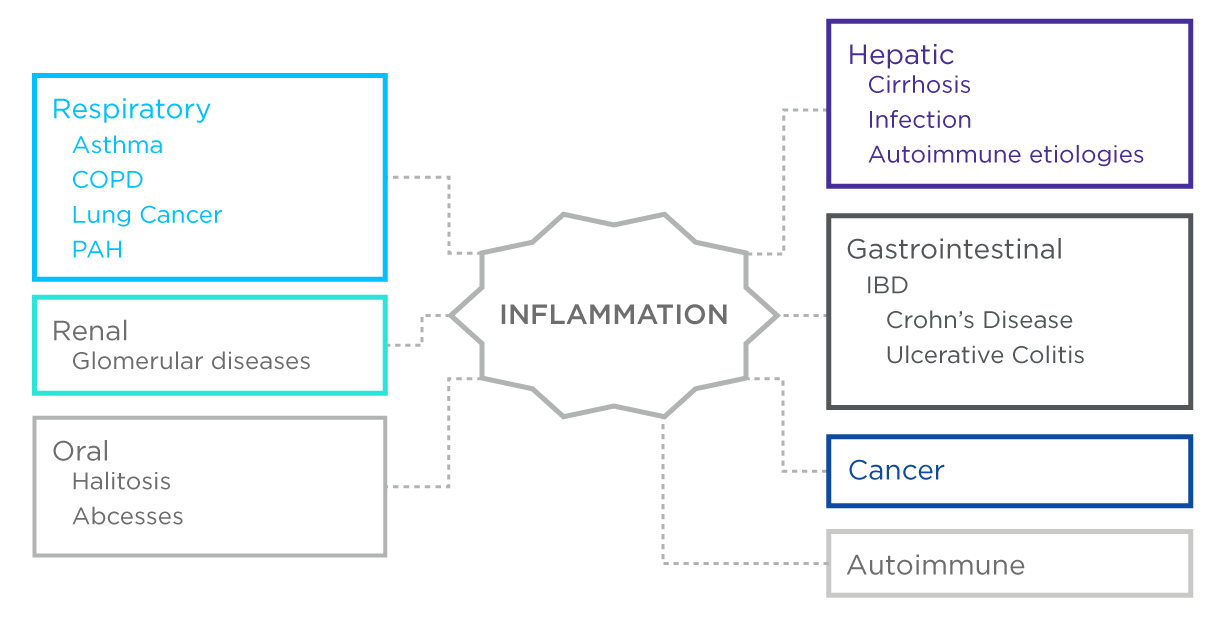
Inflammation is linked to processes including oxidative stress/lipid peroxidation, responses to hypoxia, and pathogen metabolism and breakdown. We have previously explored some of these connections in our blogs on lipid peroxidation and the Warburg effect. Notably, activated immune cells use systems associated with oxidative stress as a weapon to defend against pathogens and to eliminate damaged cells.
Damaged and dying cells are a common feature at sites of inflammation, and these too can leak distinctive macromolecules and metabolites, including the breakdown products of proteins and lipids. As such, inflammation encompasses numerous sources of potential biomarkers.
Biomarkers of inflammation can be specific
Although inflammation is a broad response seen in many illnesses, there is good reason to believe that biomarkers linked to inflammation are relevant for specific disease diagnosis and treatment.
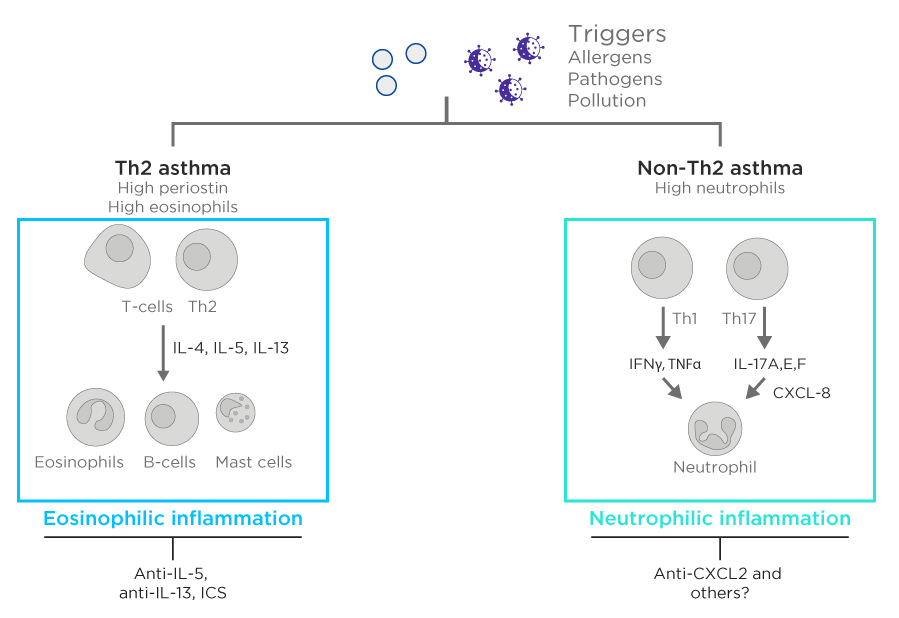
There is a great deal of redundancy in inflammation with a huge number of potential triggers that are detected and monitored by various parallel and complementary signalling systems (Figure 3), protein complexes, cell types etc. All of these converge towards a similar outcome that results in the hallmarks of inflammation. This means that there are many potential differences between seemingly similar inflammation responses and there is good reason to expect the biomarkers of inflammation to vary by disease, and even between disease phenotypes. This can be demonstrated in asthma where different inflammatory endotypes, based on the presence of different immune cells, result in distinct disease phenotypes requiring divergent treatment regimes.
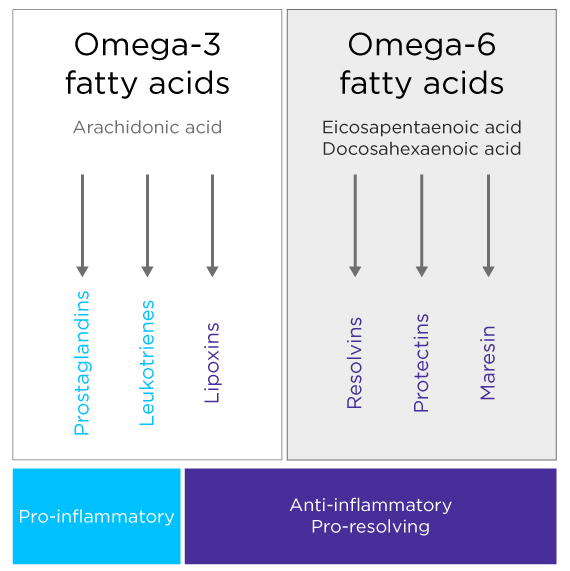
The byproducts of inflammation can also be highly context dependent (Figure 4). Processes like lipid peroxidation release small molecules as a result of the interaction of reactive oxygen species with fatty acids in cell membranes. The composition of these membranes varies between cell types, and between organelles, so it is logical to predict that there should be some relationship between the location where lipid peroxidation occurs and the cocktail of small molecules produced.
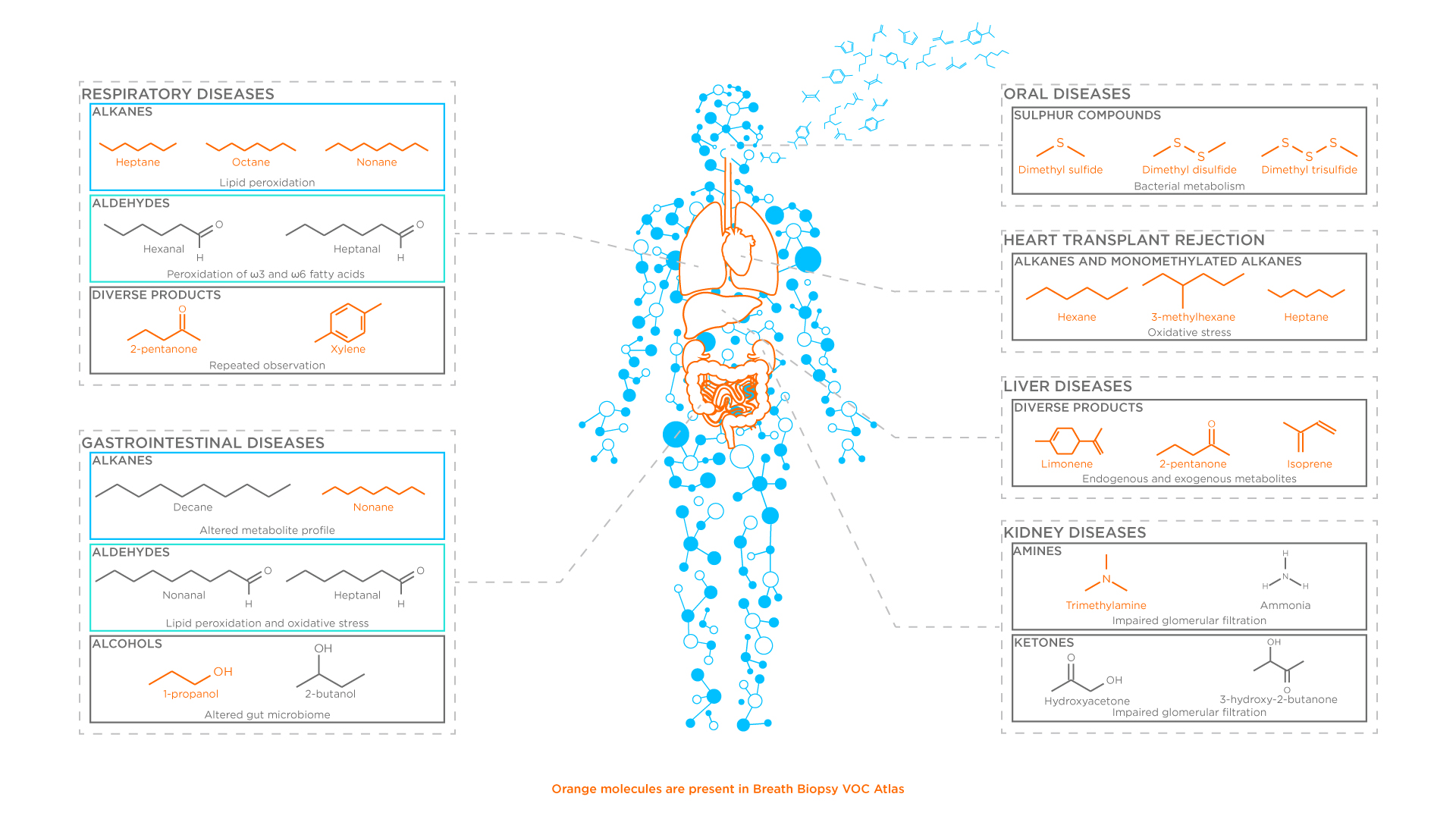
Finally, it’s unlikely that biomarkers linked to inflammation would ever be used in isolation for disease-diagnosis. Even if inflammation always produced the same complement of biomarkers, this would only form part of the diagnostic picture and it is most likely that they would be assessed alongside other biomarker research to provide a more specific result. The ability to identify cohorts of biomarkers for disease detection, depends on the application of reliable biomarker discovery tools like Breath Biopsy® OMNI, that are optimized for broad-spectrum, high-sensitivity biomarker discovery.
Still more to discover
Inflammation is a rich and varied source of prospective metabolic biomarkers, that could result in changes to VOCs on breath. Numerous studies have already proposed links between breath biomarkers and inflammation, and it is logical to predict that many others may exist. There is still a lot more to uncover about the mechanistic connections between biomarkers and biology, but the relevance of inflammation to many diseases is undeniable, and it is likely that at least some future clinical biomarkers will report on inflammatory processes.
Breath Biopsy is a reliable, non-invasive way to discover and study biomarkers on breath. If you are interested in discovering biomarkers, whether inflammatory or otherwise, talk to us about Breath Biopsy OMNI, our complete biomarker discovery service. To hear more about OMNI as well as other recent advances in breath biomarker research take a look at presentations from the Breath Biopsy Conference held on 12th and 13th October.
References
- Ibrahim, W., et al., A systematic review of the diagnostic accuracy of volatile organic compounds in airway diseases and their relation to markers of type-2 inflammation. ERJ Open Research, 2021. 7(3): p. 00030-2021. DOI: 10.1183/23120541.00030-2021
- Maskrey, B.H., et al., Mechanisms of resolution of inflammation: a focus on cardiovascular disease. Arterioscler Thromb Vasc Biol, 2011. 31(5): p. 1001-6. DOI: 10.1161/ATVBAHA.110.213850
- Durham, A.L., et al., Targeted anti-inflammatory therapeutics in asthma and chronic obstructive lung disease. Translational Research, 2016. 167(1): p. 192-203. DOI: 10.1016/j.trsl.2015.08.004
- Ratiu, I.A., et al., Volatile Organic Compounds in Exhaled Breath as Fingerprints of Lung Cancer, Asthma and COPD. Journal of Clinical Medicine, 2021. 10(1): p. 32. DOI: 10.3390%2Fjcm10010032
- Kurada, S., et al., Review article: breath analysis in inflammatory bowel diseases. Alimentary Pharmacology & Therapeutics, 2015. 41(4): p. 329-41. DOI: 10.1111/apt.13050
- Ahmed, I., et al., Investigation of faecal volatile organic metabolites as novel diagnostic biomarkers in inflammatory bowel disease. Alimentary Pharmacology & Therapeutics, 2016. 43(5): p. 596-611. DOI: 10.1111/apt.13522
- Risby, T.H. and S.S. Sehnert, Clinical application of breath biomarkers of oxidative stress status. Free Radical Biology and Medicine, 1999. 27(11): p. 1182-1192. DOI: 10.1016/s0891-5849(99)00212-9
- Ligor, T., et al., Searching for Potential Markers of Glomerulopathy in Urine by HS-SPME-GC×GC TOFMS. Molecules, 2021. 26(7): p. 1817. DOI: 10.3390%2Fmolecules26071817
- Monedeiro, F., et al., VOC Profiles of Saliva in Assessment of Halitosis and Submandibular Abscesses Using HS-SPME-GC/MS Technique. Molecules, 2019. 24(16): p. 2977. DOI: 10.3390%2Fmolecules24162977
- Phillips, M., et al., Prediction of heart transplant rejection with a breath test for markers of oxidative stress. Am J Cardiol, 2004. 94(12): p. 1593-4. DOI: 10.1016/j.amjcard.2004.08.052
