Leaders in the Field: Renelle Myers on translating breath into the clinic
Published on: 3 Aug 2022

Renelle Myers was a Keynote speaker at the 2020 Breath Biopsy Conference, presenting on ‘Breath biomarkers for the early detection of lung cancer’. Her talk is available to watch or re-watch online, at your convenience.
You’re a scientist and a medical doctor, did you always want to be both?
Medicine and science are inseparable. It’s science that moves medicine forward so that we can improve patient care.
I’m an interventional pulmonologist. My clinical field focuses on diagnosing and staging lung cancer. 80% of patients with lung cancer that need a biopsy are at an advanced stage. After giving so many life ending diagnoses, I started to think there had to be a better way – a way to find tumors earlier and improve cure rates.
I got started in the early detection of lung cancer research working with Dr. Stephen Lam. I took a position at the BC Cancer Research Institute and Vancouver General Hospital to work with his team when they were just starting a large clinical trial with lung cancer screening and low-dose CT scanning. We have taken this research and translated it to the patients’ care. We’re currently launching the first Lung Cancer Screening program in Canada. The evidence to screen for lung cancer is based on multiple large screening trials including our own, which demonstrated using risk prediction models for identifying high risk patients for lung cancer has a higher sensitivity and specificity than using age and smoking history. It’s pretty exciting to see that come to fruition after all the work.
You’ve been working on early detection and improved screening for lung cancer for a while, what led you to start looking at the diagnostic potential of breath in that context?
Low dose CT screening saves lives. It has demonstrated a lung cancer mortality reduction of up to 25 %. CT scans are highly sensitive to pick up lung nodules, but 80–90% of the lung nodules that we find are benign. It’s difficult to tell which nodules are benign unless you follow them with serial CT scans over time. We already use risk prediction models, based on smoking history, BMI and age, to try to find those high-risk individuals, but it’s not a perfect system. Ideally, we need to introduce a biomarker that can help us predict the malignant potential of the nodules so we can triage them appropriately.
For several reasons it just seemed like breath was the right place to go. It’s in direct contact with the tumor in the lungs, and it’s non-invasive and it can be sampled easily at point of care. I really do envision breath testing someday as being part of a clinician’s repertoire.
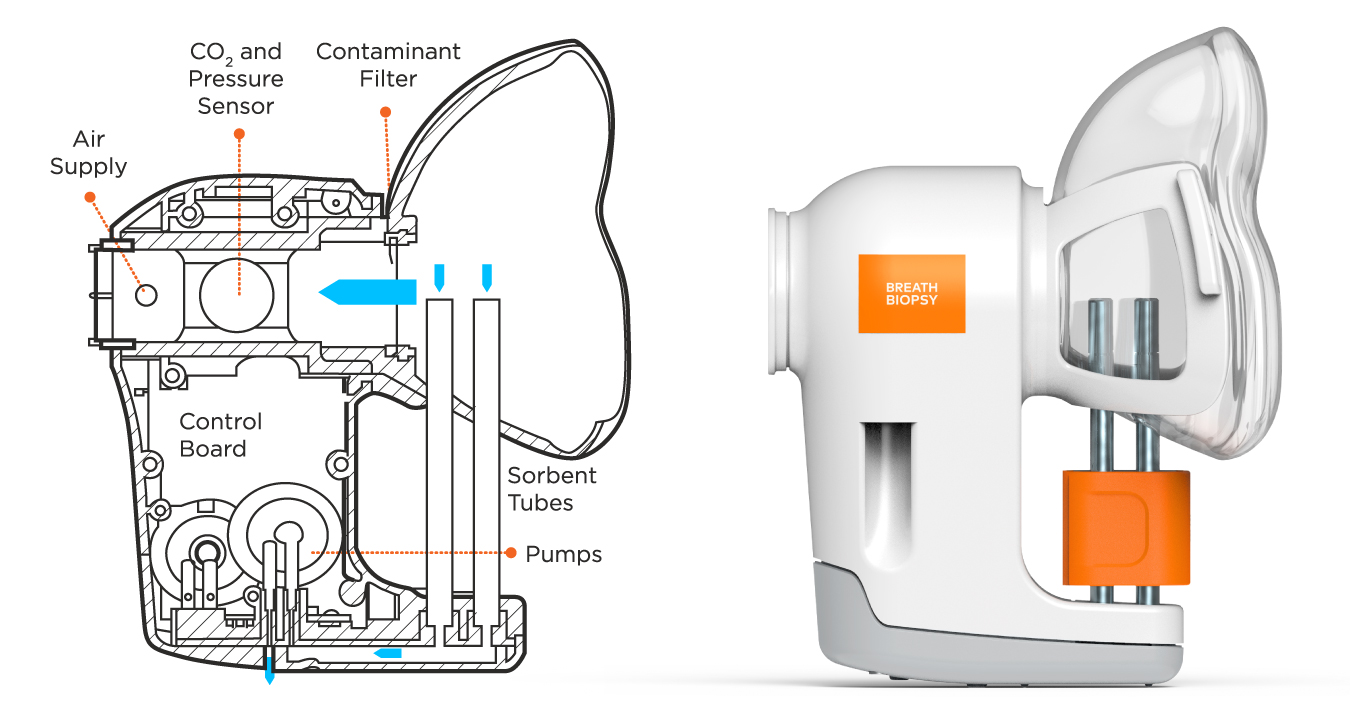
“Through the ReCIVA, Owlstone gave us a collection device that’s well designed and easy to use – it made it easier for us to enter into the realm of breath research. Since then we’ve had a lot of guidance and collaboration with Owlstone that’s really helped us get on our feet.”
I was at the World Lung Cancer Conference when I met Billy Boyle [Owlstone Medical’s CEO], and I thought Owlstone’s setup was fascinating. Breath’s an incredibly complex matrix. Analyzing it sounds like it should be easy, but every point from collection to analysis matters so much. The first breath trial we conducted was with Owlstone and we learned so much. With the ReCIVA, Owlstone gave us a collection device that’s well designed and easy to use – it made it easier for us to enter into the realm of breath research. Since then we’ve had a lot of guidance and collaboration with Owlstone that’s really helped us get on our feet.
How do you see breath tests fitting into a clinical setting in the future?
I think everyone working in breath envisions that at some point, breath is going to be a clinical tool that we can use at the point of care, whether that’s for infectious disease detection, respiratory illness classification, or monitoring disease progression. It will allow a personalized medicine approach to care. In order to develop a working test there has to be a partnership between clinicians and industry. You need the clinician saying, ‘this is the kind of test I need’, but then you need the analytic chemists, physical chemists – the people who know how to make a test that will work and give you answers at the end. Breath research is multidisciplinary.
During the pandemic, you pivoted your focus towards Covid – with your efforts getting national news coverage. How did the way you work change over the last two years?
The pandemic was difficult. All our clinical research trials were shut down for safety reasons. But it also was a blessing in disguise because, as our lab was shut down for our lung cancer research, we thought ‘Well, let’s try to detect COVID with breath’.
The WHO was saying testing was the way to control the pandemic, so we jumped on the opportunity to try and develop a breath test that would be non-invasive, point of care, rapid and hopefully accurate. A lot of people stepped away from breath because it had become a hazardous sample, but I had been working on the front line, on the respiratory wards, as a clinician, through the pandemic, so I knew what we needed to do to be safe. While it was difficult, it was also an exciting time.
Is there anything you learnt in your work on COVID you’ve been able to take back into your research in lung cancer?
We learned so much – we were a pretty new lab. We had been doing breath research, but primarily through the breath kits and analytical services from Owlstone. We set up our own lab to be able to process our own samples and there was a very steep learning curve.
We weren’t initially approved to use the ReCIVA for infectious control purposes (that’s changed now, we’re back to using ReCIVA for all our current studies). For our COVID specific study, we had to go back to using Tedlar bags. We’ve learned a lot about the limitations with those.
Now we have this huge collection of normal breath samples that we can use as controls for different trials, so it really was helpful.
You’re collaborating on a couple of exciting projects with us at Owlstone Medical at the moment, what stage is that work at?
Our work together on the early detection of lung cancer was derailed by COVID. We were looking at patients who had lung cancer then had a resection, and we were using repeat breath samples to see if we could we see a difference in that signal post operative when the tumor has been removed. The bulk of that has been done and I do think that’ll be published very soon.
The other exciting thing we’re doing with Owlstone is looking at the effects of air pollution on healthy controls. In British Columbia, we have the Air Pollution Exposure Lab (APEL), so in partnership with Owlstone, we’ve been doing exposures on healthy controls, and then collecting serial breath samples to see if we get an influx of inflammatory breath biomarkers. We need to know what is actually happening if we want to try and prevent that inflammatory response. That’s going on currently and we should be done with recruitment by the end of August.
How can we know if a VOC we’re measuring is actually on breath, rather than an ambient contaminant?
“Meticulous environmental sampling is really important. Every breath collection that we do has a paired environmental sample.”
This is one of the major problems that remains in breath. Meticulous environmental sampling is really important. Every breath collection that we do has a paired environmental sample. But, when you’re taking 1000s of samples, one-to-one background samples can mean literally months of extra processing time. It’s never going to be 100% but using a system like ReCIVA with the CASPER clean air supply that removes a lot of contaminants at the sample collection stage is a big help in that area. Then it comes down to having really careful background evaluation.
However, once you know whether a VOC is on breath there are still many questions. How do you know a VOC is from the lungs and not gastrointestinal? Even the mouth is going to have a different VOC profile than the alveolar profile. I think we have to get away from the thought that there’s going to be just one or two molecular features that are diagnostic.
What can the breath research community do collectively to ease the path towards potential breakthroughs?
“If we’re going to develop a clinically useful biomarker for many clinical diseases, we need to acknowledge that no one team or lab is going to do that.”
Breath research is hard. There’s always something that’s going to derail you. I found as a clinician trying to work through COVID that we had to turn breath into an agile science. That was difficult because breath also has to be meticulous, but we found a balance and I think that’s what we need to do going forward to capture those clinical opportunities.
In breath you often see studies looking at just 20-40 people for biomarker discovery work, but we need to see the breath world coming together to run large international multicenter, multicultural studies in different areas of the world, looking at the same things. We need to standardize the breath workflow from collection to data processing so the same methods can be applied around the world and data collection will be a global effort. There’s a lot of great work with the peppermint protocol that looks at quality control across multiple centers and machines and devices. That’s what breath needs before a clinician is going to adopt it into their clinical practice.
If we’re going to develop a clinically useful biomarker for many clinical diseases, we need to acknowledge that no one team or lab is going to do that. There are so many great researchers, but we need to work together on it.
Recent papers from Renelle Myers:
- High-Ambient Air Pollution Exposure Among Never Smokers Versus Ever Smokers With Lung Cancer
- Breath collection protocol for SARS-CoV-2 testing in an ambulatory setting
- Peppermint protocol: first results for gas chromatography-ion mobility spectrometry
- USPSTF2013 versus PLCOm2012 lung cancer screening eligibility criteria (International Lung Screening Trial): interim analysis of a prospective cohort study
This blog post is part of our ongoing series of in-depth interviews with influential leaders currently working in breath research. Other ‘Leaders in the Field’ interviewees have included Professor Richard Yost, inventor of the triple quadrupole mass spectrometer, Salman Siddiqui, a specialist in breath testing for inflammatory respiratory diseases, and Anke-Hilse Maitland-van der Zee, President of the Federation of Innovative Medicine Research in the Netherlands.
READ MORE INTERVIEWS IN THIS SERIES
Renelle Myers’ Keynote talk from #BBCon20 is available to watch on demand, alongside all of the other presentations from previous Breath Biopsy Conferences. There’s still time to register for #BBCon22 and join with influential voices in breath research, this November.