Breath Biopsy: Robust and Reproducible Breath Analysis
Published on: 29 Nov 2017
Exhaled breath contains thousands of volatile organic compounds (VOCs), valuable biomarkers that reflect metabolic processes taking place within a person’s cells and tissues. Measuring VOC biomarkers in breath could offer a completely non-invasive way of monitoring health and diagnosing disease. However, robust breath biomarker discovery requires highly reproducible workflows for breath collection and VOC analysis.
In the past, a high variability between breath sampling techniques has resulted in a lack of reproducible results1,2. The ReCIVA® Breath Sampler, part of Owlstone Medical’s Breath Biopsy® platform, was designed in collaboration with experts in the breathomics field to provide a standardized method to collect exhaled breath samples. By sampling onto sorbent tubes, ReCIVA overcomes the challenges associated with collecting breath in containers such as Tedlar® bags, which have been shown to result in the loss of sampled VOCs.3

Repeatable Breath Sampling
Reproducibility between breath collects ensure comparability between samples and patients. It is particularly important when making longitudinal measurements of VOC biomarkers, where it is vital to capture accurately the real changes in biomarker concentration over time.
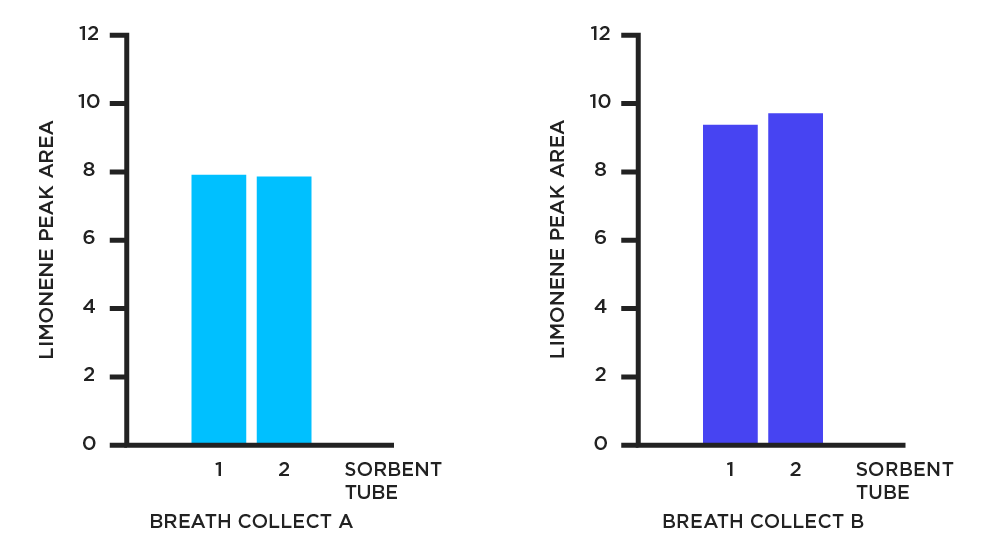
Data from our Breath Biopsy laboratory demonstrates the robustness of the breath sampling and analysis workflow, including breath collection using the ReCIVA Breath Sampler and VOC analysis using FAIMS (field asymmetric ion mobility spectrometry). Figure 2 shows measurement of limonene in breath collected in duplicate on two sorbent tubes during two separate breath collects (A and B) from the same individual.
FAIMS – excellent reproducibility for VOC analysis
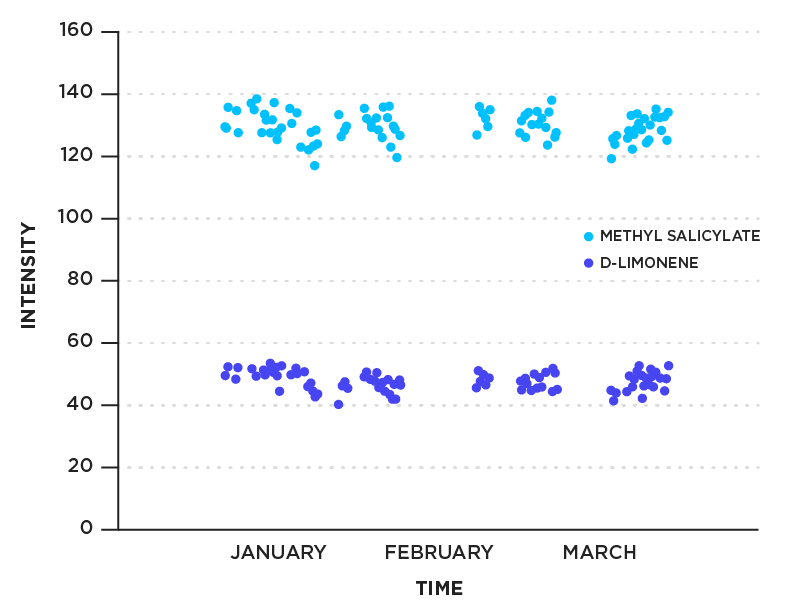
The Breath Biopsy platform uses FAIMS to analyse breath samples. To demonstrate FAIMS reproducibility, known quantities of methyl salicylate and limonene standards were loaded onto sorbent tubes and analyzed using GC-FAIMS (Lonestar VOC Analyzer). The experiment was repeated regularly to assess the variation over time (Figure 3). Data for the two compounds over a three month period are shown. The relative standard deviation is 6.1 % for methyl salicylate and 3.4 % for limonene.
Examples of longitudinal measurements of breath VOCs in the scientific literature – READ MORE
Breath Biopsy®
If you want to learn more about Owlstone Medical’s Breath Sampling and Analysis technology, and our Biomarker Services, why not download our free ebook: Breath Biopsy: The Complete Guide?
References
- Risby, Critical issues for breath analysis, J Breath Res, 2008, PDF of article
- Herbig and Beauchamp, Towards standardization in the analysis of breath gas volatiles, J Breath Res, 2014, https://doi.org/10.1088/1752-7155/8/3/037101
- Beauchamp et al., On the use of Tedlar® bags for breath-gas sampling and analysis, J Breath Res, 2008, https://doi.org/10.1088/1752-7155/2/4/046001

