OMNI: Reproducible Non-invasive Global VOC Analysis of Breath
Breath Biopsy® OMNI provides unrivalled capability for non-invasive global VOC analysis, with a focus on reproducible measurement of a broad range of biorelevant compounds on breath. As a complete end-to-end service, OMNI encompasses sample collection, processing and data analysis as well as access to our established expertise in study design, clinical trial management and data interpretation.
Published on: 29 Nov 2021
The huge potential of non-invasive biomarkers on breath means that hundreds of studies have been published investigating breath in a wide range of biological situations from cancers, respiratory and cardiometabolic diseases to workplace exposures, drug detection and infections. These studies use a vast selection of approaches to collect and analyze volatile organic compounds (VOCs) on breath and, as such, comparison between studies is currently a challenge.
Breath Biopsy® OMNI® provides unrivalled capability for non-invasive global VOC analysis, with a focus on reproducible measurement of a broad range of biorelevant compounds on breath. As a complete end-to-end service, OMNI encompasses sample collection, processing and data analysis as well as access to our established expertise in study design, clinical trial management and data interpretation.
This blog summarises just some of the work we’ve done to develop OMNI to provide reliable, reproducible breath analysis.
What does OMNI do?
OMNI has been developed and optimized to identify and measure VOCs on breath. Many breath research studies report the total number of signals detected from the analysis of a breath sample. While some of these may be important, there are also many other potential sources for signals including ambient contamination and sampling hardware related contaminants, which can contribute to high rates of false discovery.
|
We define a compound as being ‘on breath’ when the area under the curve in a breath sample is greater than three standard deviations above the average abundance in blank samples. |
In developing OMNI, we compared many breath samples to various blank control samples allowing us to better identify signals that occur on-breath. We have developed a measure of confidence for differentiating VOCs mainly arising in the breath matrix, from those with other sources. This method helps to ensure that a compound will only be considered in our analysis if it occurs on breath at significantly higher levels that in blanks (Figure 1). You can find out more about this in a previous blog. On-breath signals are more likely to relate to specific VOCs and to have connections to biological and disease processes. By distinguishing on-breath compounds in this way OMNI reduces false discovery and increases the chance of finding clinically relevant biomarkers.
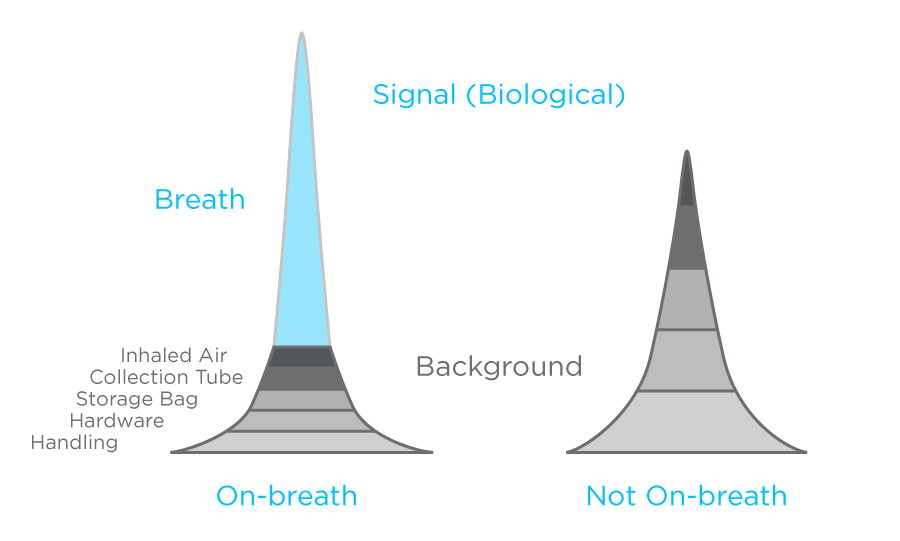
Figure 1: Representations of mass spectrometry peaks and the possible sources of the molecules responsible for each peak. While some peaks represent componds that largely originate on breath, others may not.
What makes OMNI special?
OMNI is the product of years of internal research to develop a breath biomarker pipeline that is robust, reliable and offers high quality results. We have examined, reviewed and improved technologies and processes throughout the pipeline resulting in a system that can reliably detect up to 500 VOCs on breath. Here are just some examples of the distinctive capabilities of OMNI:
- Expertise at every stage – Owlstone Medical have over 15 years of experience in trace compound detection and have spent more than five years investigating breath as a source of clinically-relevant biomarkers. Our team can provide specialist support in study planning & design, clinical trial management, statistical analysis of results, and biological interpretation of findings. All of this is available as part of the OMNI service.
- Selective breath sampling – Unless otherwise required, OMNI breath samples exclude air from anatomical dead space in the upper airways. This means that Breath Biopsy samples mainly include exhaled breath from gas exchange rich areas of the lungs, enriching for VOCs exchanging from the blood. The ability to selectively collect breath fractions during passive breathing is a key feature of the ReCIVA® Breath Sampler and enhances biomarker detection by excluding air that has not been exposed to the bloodstream. ReCIVA is able to do this because its sensors constantly measure and monitor breathing patterns.

- Consistent and reproducible analysis – Our analytical process includes a thorough series of quality assurance stages. To validate this, we include consistent quantities of over 50 standard compounds in every sample analytical sequence. The standards have been selected to represent a sample of the diversity of relevant compounds. In addition, eight deuterated standards are included in each sample, allowing for a sample-by-sample assessment of analytical performance. Including such an extensive range of samples ensures a fit for purpose assessment of the quality of the results from each sequence and allows us to spot any issues arising during sample processing.
- Enhanced feature extraction – We employ customized software settings to ensure more consistent identification of molecular features from the results of mass spectrometry analysis. Our optimization efforts mean that 98% of signals from standard VOCs are correctly detected by OMNI, and OMNI is able to detect over 70% more compounds on breath due to enhanced feature extraction, compared to an equivalent out-of-the-box feature extraction process. Overall, these improvements more than double the number of prospective breath biomarkers consistently detected by OMNI.
How can OMNI help you?
OMNI provides the most robust, rigorously tested method available for non-invasive breath biomarker analysis. If you’re seeking a way to identify and develop biomarkers that are clinically relevant and can be collected in a wide range of settings including at home and in primary care then ask us more about OMNI.
We offer a range of analysis reporting options depending on your needs including univariate and multivariate statistics, and biological interpretation to propose relationships between identified compounds and relevant biological processes. OMNI results are prepared to provide a clear route to compound identification facilitated by our bespoke Breath Biopsy HRAM Library of chemical standards. Choosing to use a specialist, standardized process like OMNI facilitates performance of replication studies, a vital aspect of helping to validate and progress biomarkers towards clinical endpoints.
OMNI is suitable for investigating a wide range of clinically relevant research questions and we can help you to design your study to provide quality results in line with your resources and requirements. Visit our product page to find out more about what OMNI has to offer and watch our recent presentation along with many others at the Breath Biopsy Conference 2021 for a more in-depth look at the technology behind OMNI. If you are interested in reliable VOC analysis for biomarker studies and would like to add OMNI to your research, please get in touch and our team will be happy to discuss your needs.
