Unlock the secrets of the gut microbiome with breath testing
Published on: 13 Dec 2022
The last twenty years have seen significant growth in both breath research and research into the human microbiome. The recent overlap of these fields has been especially beneficial and has the potential to usher in a new era of personalized medicine.
Much of the progress in breath research over recent years has focused on the identification of volatile organic compounds (VOCs) on exhaled breath. Breath VOCs can originate either from within the body (endogenous VOCs) or from external sources such as diet, prescription drugs and environmental exposure (exogenous VOCs). Endogenous VOCs are often products of metabolic activity in the body and changes to these VOCs can be characteristic of specific disease processes. But many of the metabolites made within the human body are not actually the products of human metabolism, they are created by the metabolism of the countless microbes that thrive within and enrich our bodies.
Trillions of microbes live within the human body, with perhaps the highest concentrations to be found within our gut where we rely upon them to symbiotically help us digest the food we’ve eaten. Our gut microbiome has a huge role to play in establishing and maintaining good health.
We’re incapable of digesting much of the fibrous material we eat as part of a normal diet – we don’t naturally produce the enzymes we need to break these fibers down. Established tests that evaluate the health of our gut microbiome have been relatively invasive and/or unpleasant (stool samples, endoscopies, blood tests, lumen swabs, etc). However exhaled breath is an unlimited, easy-to-collect, and comparatively comfortable testing option, and recent research suggests that changes in gut bacteria and health are mirrored on our breath.
What can we see on breath?
The volatile nature of many of the bacterially derived metabolites make the microbiome ideal for analysis via breath testing. A recent study from Smolinska et al. found that VOCs on breath relate either directly or indirectly to processes within the gut microbiome, in a way that was sensitive to the changes in active and inactive gastrointestinal disease states.1 Additionally, breath can provide you with a dynamic real time insight into the health of the gut biome. In contrast, gut biome testing via fecal analysis is more reflective of what was happening in the gut as long as 48 hours ago.
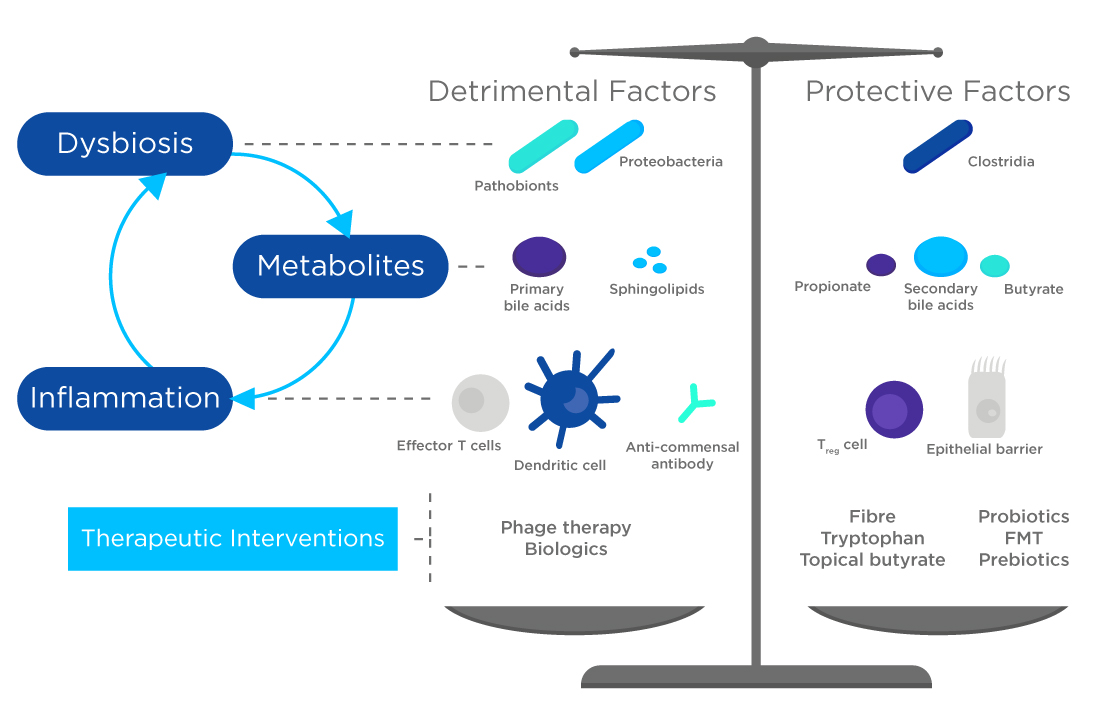
Certain food ingredients present in the large intestine, lead to the production of various volatile metabolites including short chain fatty acids (SCFAs) such as acetate, butyrate, and propionate, but also alcohols such as ethanol and propanol. These bacterial fermentation products are detected in exhaled breath, and have a range of beneficial effects including anti-inflammatory, anti-oxidative and anti-carcinogenic potential and are associated with a decreased risk of developing cancer, gastrointestinal disorders, and cardiovascular disease.
Aromatic amino acid catabolism by the gut microbiome also yields numerous metabolites that may regulate immune, metabolic, and neuronal responses at local and distant sites. Volatile bacterial metabolites that are products of aromatic amino acid catabolism (including phenol, phenyl acetaldehyde, p-cresol, and others) could also be analysed via a gut health focused breath test.
At Owlstone we’re conducting work, alongside our partners in industry, to progress the field and increase understanding of the gut microbiome and its relationship with exhaled breath. We conducted microbiome anaysis as part of our recent studies showed significant changes in some VOCs before and after iron supplementation and at various time points post treatment for iron deficiency anemia. Some SCFAs, butanoic, propanoic and acetic acids, significantly increased after 28 days of iron supplementation – metabolites previously linked to increased gut health – and there was a concomitant decrease in hydrogen sulfide, a compound linked to poor gut health.
We can help you investigate the gut microbiome
As interdisciplinary specialists in breath, we have created Breath Biopsy® OMNI® to be the most advanced solution for reliable global breath biomarker analysis. And, when you choose to work with us, your investigation is further supported by our extensive study design, management, and data interpretation expertise.
When you work with us you also get access to our Breath Biopsy VOC Atlas. The VOC Atlas is a comprehensive catalogue of VOCs identified on exhaled human breath that will facilitate future biomarker discovery and can be used as a comparison in multiple disease contexts and analysis methods to characterize relationships between the gut microbiome and breath VOCs. Currently more than 150 VOCs are registered within the ATLAS, having been rigorously identified and confirmed as found in statistically significantly higher concentrations on breath as opposed to within the ambient background. Gut microbiome-associated VOCs already added to the ATLAS include sulphurous compounds, short chain fatty acids (SCFAs), and aromatic amino acid breakdown products.
Watch breath Biopsy OMNi webinar READ More on the VOC Atlas
Hydrogen and methane are already established breath testing biomarkers, exclusive to the gut microbiome, that are used to characterize gut disease. They are produced by bacterial / archaeal metabolism that can be indicative of dysbiosis, food intolerances or digestive issues, such as small intestinal bacterial overgrowth or inflammatory bowel disease. While hydrogen and methane are not compatible with our Breath Biopsy OMNI platform we can offer a complementary analytical method that will allow you to fully characterize these established VOC biomarkers of the gut microbiome on exhaled breath.
Talk to us about hydrogen and methane breath testing
References
- A. Smolinska et al. Volatile metabolites in breath strongly correlate with gut microbiome in CD patients, Analytica Chimica Acta. (2020) 1025: pp 1-11. DOI: 10.1016/j.aca.2018.03.046
- Lavelle A, Sokol H. Gut microbiota-derived metabolites as key actors in inflammatory bowel disease. Nature Reviews Gastroenterology & Hepatology. (2020) 17(4): pp223-237. DOI: 10.1038/s41575-019-0258-z
