Using breath to diagnose invasive fungal diseases
Non-invasive testing would be a huge advantage for vulnerable populations
| Publication information: S. Koo et al. A Breath Fungal Secondary Metabolite Signature to Diagnose Invasive Aspergillosis Clin Infect Dis. 2014, 59(12): 1733-1740. DOI: 10.1093/cid/ciu725
Disease Area: Aspergillosis, Infectious Diseases, Respiratory Disease Application: Diagnostics Sample medium: Breath Analysis approach: TD-GC-MS Summary:
|
Invasive aspergillosis (IA) is a particularly dangerous infection, associated with stubbornly high mortality rates (25%-58%) in immunocompromised patients, despite recent advances in antifungal therapies. These mortality rates are at least in part attributable to the current difficulty in achieving an accurate and early diagnosis.
The symptoms associated with IA are nonspecific and even radiological testing has difficulty separating IA from other invasive fungal diseases. The existing samples that can be used to diagnose this infection all have their drawbacks – with serum and bronchoalveolar lavage currently the most common. The process for acquiring a bronchoalveolar lavage sample is invasive and unpleasant and lacks the ideals levels of sensitivity. Serum is slightly less invasive but even less sensitive. A tissue biopsy would have a much higher sensitivity than either of these options. However, tissue is not an ideal option for the already debilitated patients that IA most commonly effects, as it’s the most invasive sample of the three.
It is clear that if a non-invasive breath test could be developed that was capable of accurately diagnosing IA infection it would be of huge benefit. There is reasonable cause to expect breath tests could be well suited to detecting aspergillosis. Pathogens such as bacteria and fungi have metabolic pathways that are distinct from those normally present in humans. Many of the metabolites produced by IA fungi in the lungs are volatile, and so could potentially, once identified, be easily collected and analyzed on breath.
For their study, Koo et al. attempted to identify volatile organic compounds (VOCs) associated with growth of IA in vitro then examined, whether they could find evidence of the same compounds occurring in vivo, on the breath of patients being evaluated for IA infection.
Methods
Breath samples were collected on desorption tubes from 64 patients with suspected invasive fungal pneumonia. These patients were then classified as either true IA cases (34) or non-IA cases (30) independently by two investigators according to the current standards for diagnostic classification for patients with invasive fungal diseases.
The breath samples were analyzed via TD-GC-MS, with molecular features of interest identified provisionally using the NIST Library. Key metabolites were further verified with pure chemical standards.
Results
Of the 64 patients sampled, 34 were diagnosed with IA and 30 with other forms of pneumonia. ‘There were no adverse events related to the breath collection procedure; it was well tolerated, even by patients who were dyspneic or requiring supplemental oxygen.’ A diagnostic model based upon the metabolites β-trans-bergamotene, α-trans-bergamotene, trans-geranylacetone and a β-vatirenene–like sesquiterpene distinguished the breath of patients with and without IA with 94% sensitivity and 93% specificity.
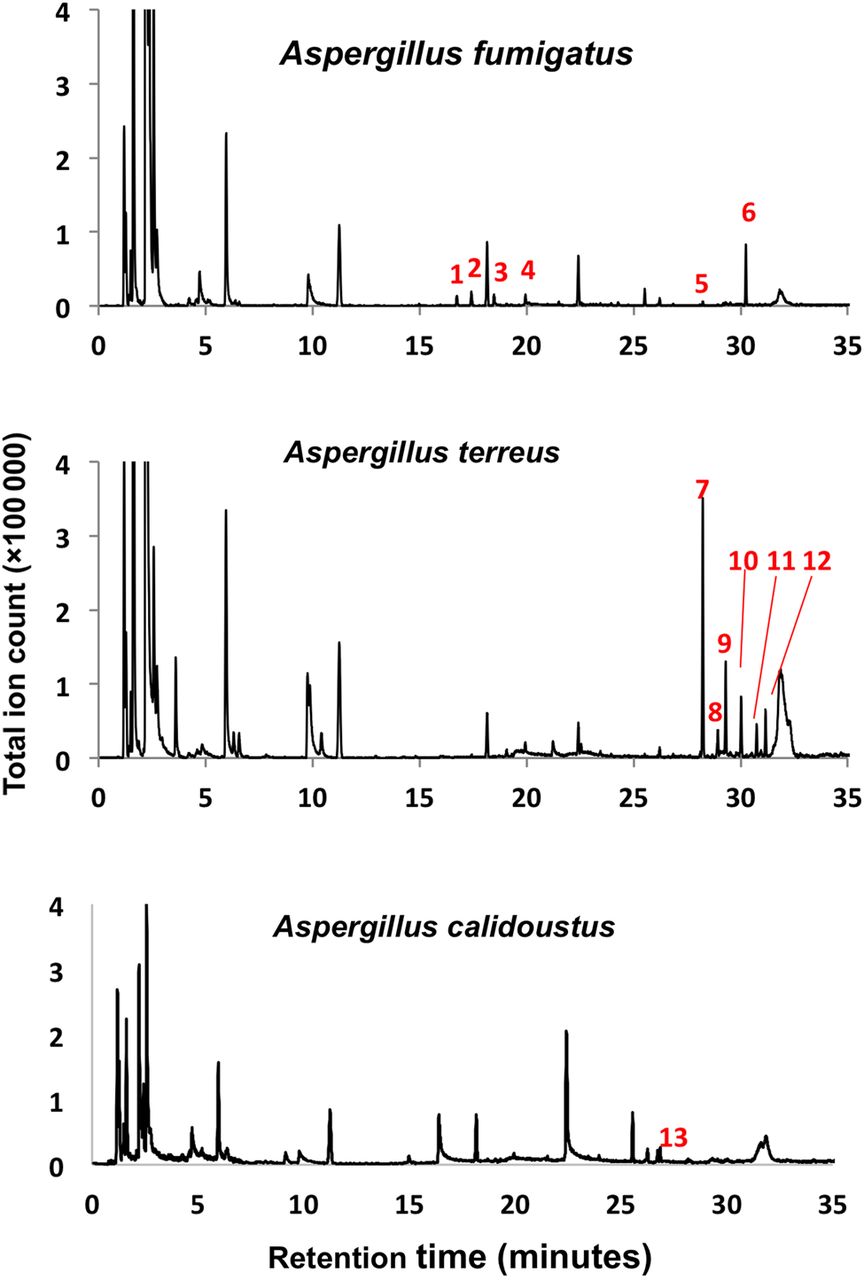
Figure 1: In vitro volatile organic compound profiles of Aspergillus fumigatus, Aspergillus terreus, and Aspergillus calidoustus. Aspergillus species have distinctive volatile organic compound profiles, with particular interspecies heterogeneity in monoterpene and sesquiterpene metabolites. The following peaks were identified as (1) α-pinene; (2) β-pinene; (3) camphene; (4) limonene; (5) α-trans-bergamotene; (6) β-trans-bergamotene; (7) elixene; (8) santalene; (9) elemene; (10) acoradien; (11) 1,5,9-trimethyl-1,5,9-cyclododecatriene; (12) chamigrene; and (13) β-sesquiphellandrene.
These results suggest that, with further investigation, ‘direct detection of exogenous fungal metabolites in breath can be used as a novel, noninvasive, species-specific approach to identify patients with IA, potentially allowing more precise targeting of antifungal therapy and fewer invasive diagnostic procedures.’
Conclusions
Koo et al. proposed biomarkers for IA based on in vitro work that attempted to identify distinctive secondary metabolites of the most common pathogenic Aspergillus species. Koo et al.’s biologically guided approach relies upon the fact that pathogens, such as Aspergillus, may be producing unusual VOCs not normally produced via healthy human metabolism and thus not normally found on human breath. This approach successfully led to finding a set of four fungal compounds in human breath that could identify IA with high accuracy.
Further work in this area, beyond validation of the proposed VOCs, might add value by looking for specific prognostic indicators or focusing on the relationship between severity of infection and abundance of the key VOCs, or attempting to identify distinguishing VOC features for other less common fungal species.
If you would like to conduct a breath study in this area, or a related one, you might be interested in reading our top tips for designing an effective breath study, or browsing our collated resources for using Breath Biopsy® to study respiratory diseases.
TOP STUDY DESIGN TIPS BREATH BIOPSY FOR RESPIRATORY DISEASES
To talk to our experts about starting a study using Breath Biopsy get in touch.
Breath Biopsy offers complete support for sample collection and analysis. Find out about our analysis services