Services
Combining high resolution accurate mass mass spectrometry and world-leading breath analysis expertise to offer complete biomarker discovery and validation solutions
INTRODUCTION
Enabling early disease detection and precision medicine across a wide range of disease areas
Through Breath Biopsy® Services we can support every aspect of your breath biomarker research. From trial design through to analysis, test development and validation, our experienced team can help. We recommend accessing our services as part of Breath Biopsy OMNI, our most advanced solution for reliable on-breath global biomarker discovery including optimized methods for sample collection and analysis.
As a user of Breath Biopsy Services, all of your breath sample processing and analysis will be handled by our expert team at the world’s only specialist high-volume Breath Biopsy Laboratory in Cambridge, UK.
Our analytical pipeline centers on high resolution accurate mass (HRAM) mass spectrometry (MS) provided by Q Exactive™ Orbitrap™ technology and includes rigorous quality control at all stages to ensure you get the best possible results. Biomarker identification uses the optimized HRAM Breath Biopsy Library and our in-house Breath Biopsy VOC Atlas to give high confidence molecular identities.
Breath Biopsy Products and Services are for research use only. Not for use in diagnostic procedures.
Analytical Services
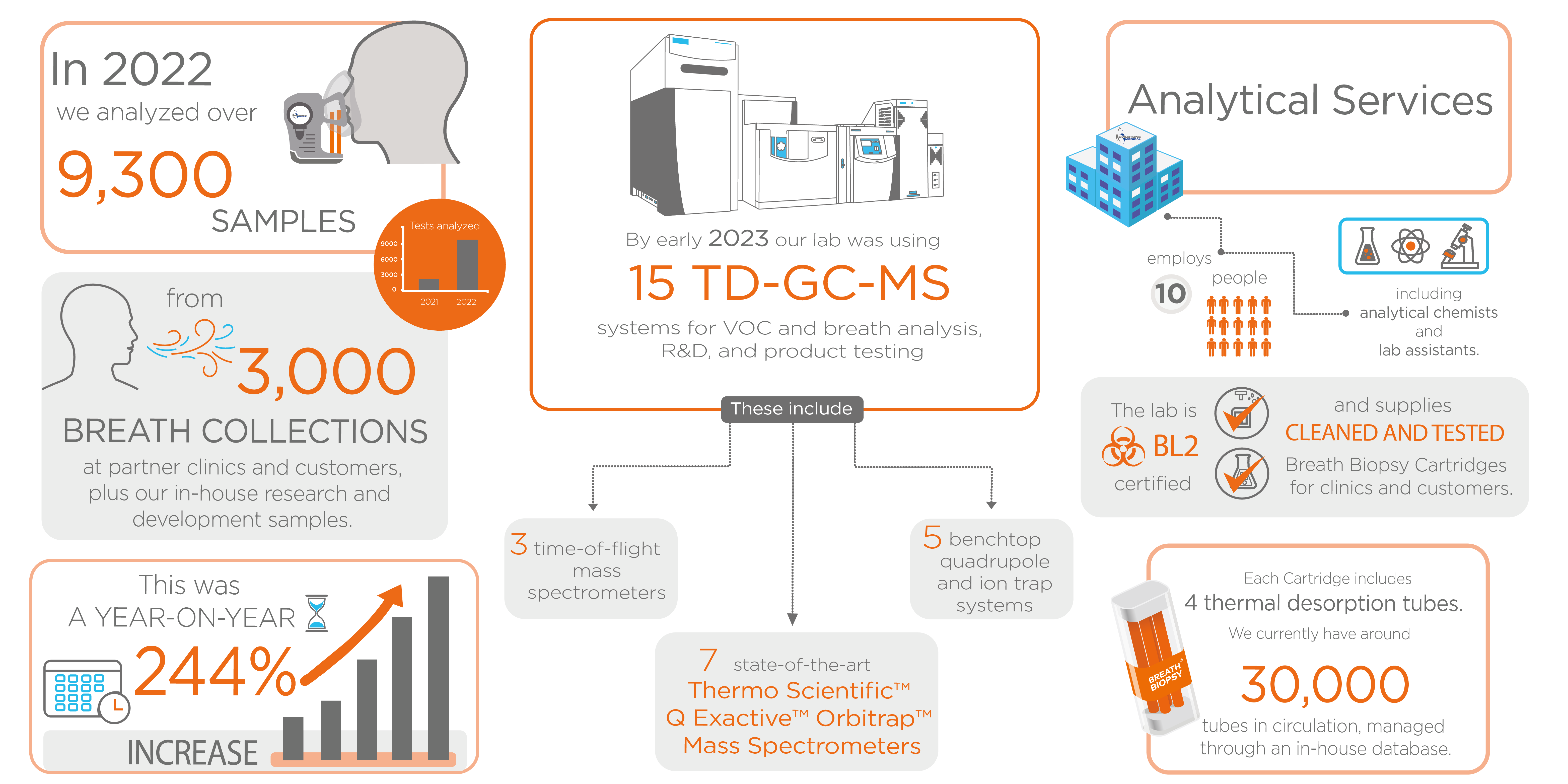
Service Capabilities
- SENSITIVITY
High sensitivity prospective breath biomarker detection
- VERSATILITY
Service options available for focused and VOC discovery analysis to suit your needs
- PRECISION
HRAM mass spectrometry VOC analysis
- CUSTOMIZATION
Data processing options to include in-depth statistical analyses and modelling
- ACCURACY
Tailored HRAM Breath Biopsy Library of VOCs and Breath Biopsy VOC ATLAS provides confidence in identification
- DISEASE RELEVANT
Prospective biomarkers with reported relevance to a range of key disease processes, including inflammation
- QUALITY
Rigorous quality control standards
- INTERPRETATION
Biological interpretation and relevance analysis
Breath Biopsy OMNI
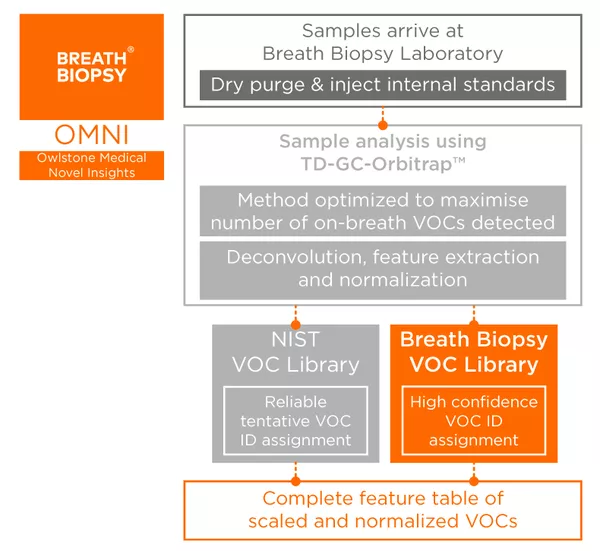
By optimizing breath collection and analysis, we have developed Breath Biopsy OMNI, our most advanced solution for reliable on-breath global biomarker discovery. OMNI has been developed to maximize compound detection and minimize process variability, ensuring high-confidence biomarker discovery that allows rapid compound identification.
Our methods, specifically assess on-breath compounds, helping to verify that detected compounds have biological relevance and are not simply reflective of ambient factors. Our HRAM Breath Biopsy Library, coupled with our Breath Biopsy VOC Atlas, also offers high-confidence prospective identification for many on-breath compounds.
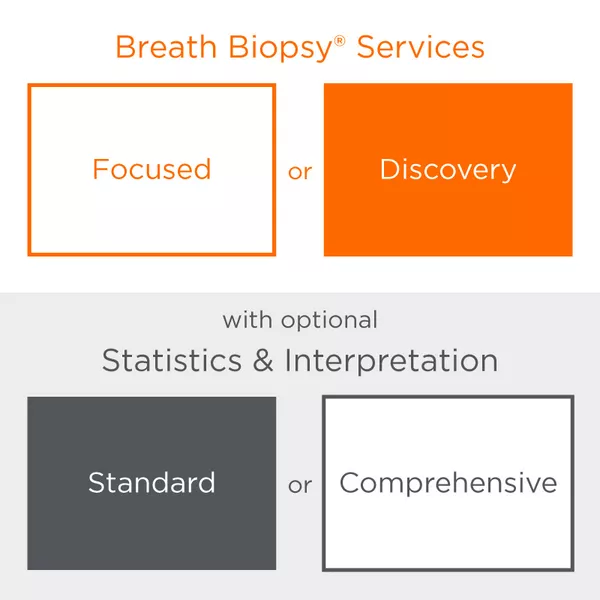
Your Breath Biopsy Service Options
Every breath research project is different and has its own set of needs and requirements. Our Breath Biopsy Services can be adapted to suit the specifics of your study, reflecting your field of interest, resources, budget and access to expertise.
Breath Biopsy OMNI is our complete solution for global biomarker discovery, including comprehensive analysis and VOC identification with high-confidence identify assignments matched from the HRAM Breath Biopsy and NIST libraries.
Results include a raw feature table with relative quantifications of all analyzed compounds. Should you wish, you can also request additional data analysis and reporting for your study. Reporting is provided at two levels, Standard and Comprehensive, and includes statistical analyses, modelling and biological interpretation. Talk to our team to find out which option best suits your needs.

HRAM Breath Biopsy Library
The HRAM Breath Biopsy Library provides reliable breath biomarker identification optimized for Breath Biopsy samples. VOCs included in the Library have relevance to factors including inflammation, oxidative stress, respiratory disease, cancer, liver disease, the microbiome or environmental exposures. Each association is supported by a combination of biological rationale, mechanistic relationships and strong empirical evidence. Currently there are more than 400 VOCs in our HRAM Library.
Breath Biopsy VOC Atlas
The Breath Biopsy® VOC Atlas is a catalogue of identified and quantified VOCs that are commonly found on breath within a heterogenous healthy population. It currently contains more than 150 VOCs. We created the Atlas to provide insight and scientific context to these molecules with the goal of enabling confident selection of candidate biomarkers for disease. The Breath Biopsy VOC Atlas is part of the OMNI platform and allows us to identify VOCs on breath with a higher degree of confidence, further enhancing our ability to understand the underpinning biology and validate non-invasive biomarkers.
RUO Panel for Respiratory Diseases
We developed our Panel to drive meaningful advances in biomarker research on respiratory diseases. Quality biomarker research requires high confidence biomarker identification and clear evidence of biological relevance. The Breath Biopsy Panel for Respiratory Diseases consists of a curated set of VOC biomarkers on breath that can be reliably identified through the HRAM Breath Biopsy Library and that show demonstrable associations to respiratory diseases and disease processes.
The Panel can support research to characterize disease endotypes, provide disease monitoring and measure therapeutic responses. Panel VOCs can be analyzed with our focused or discovery Service options of the OMNI Assay. We also provide a range of options for data interpretation and reporting to fit the needs and requirements of your study.
Find out about the specific VOCs included in the panel by downloading our poster:
Breath Biopsy offers complete support for sample collection and analysis. Find out about our analysis services
Orbitrap for Breath Biopsy
- OPTIMIZED
To enable high-quality VOC detection from breath samples
- SENSITIVITY
Capable of detecting low abundance analytes in a complex breath matrix
- DYNAMIC RANGE
Detect both high-abundance VOCs and trace-level analytes within a single analysis. Typical concentrations of chemicals in breath span 5 orders of magnitude.
- RELIABLE
Accurate mass stability and spectral fidelity leads to fast and confident detection.
- SOFTWARE
Software flexibility for discovery and routine targeted workflows.
With you at every stage
As the leaders in Breath Biopsy, we’re equipped to help you at every stage of your investigations. Thanks to our role in some of the world’s largest breath research trials, our team includes authorities on study design and planning. We offer study management and training to implement your studies in clinical sites worldwide.
Developing clinical applications
Beyond biomarker discovery and validation, we can work with you to develop identified biomarkers into focused breath-based tests for clinical implementation. We have the capabilities to work with you to develop your research further through fit-for-purpose quantitative TD-GC-MS assays or our proprietary field asymmetric ion mobility spectroscopy (FAIMS) solutions.
FAIMS combines high sensitivity, tuneable chemical detection with versatility and portability making it suitable for application in a range of point of care clinical settings. Together, we can develop tests for use in screening, early detection, complementary diagnostics, treatment monitoring and much more.