Differentiating asthma inflammatory phenotypes via breath
Breath offers the possibility of rapid and non-invasive precision medicine
| Publication information: Florence N. Schleich et al. Exhaled Volatile Organic Compounds Are Able to Discriminate between Neutrophilic and Eosinophilic Asthma, American Journal of Respiratory and Critical Care Medicine, 2019; https://doi.org/10.1164/rccm.201811-2210OC
Disease Area: Asthma Application: Patient stratification Sample medium: Breath Analysis approach: GC-MS Compounds identified in this study are available for focused, high confidence analysis through the Breath Biopsy RUO Panel for Respiratory Diseases. Summary:
|
An estimated 339 million people worldwide have ‘asthma’1 – a general term that is used to refer to a broad spectrum of conditions that cause chronic inflammation in the airways of the lungs. A lack of accurate stratifying diagnostics means that treatments are often prescribed via ‘trial and error’ approach.
Prescription of inappropriate treatments is unnecessary spending for the healthcare provider and leads to poor disease control and increased risk of exacerbations for the patient. Breath analysis offers the possibility of a rapid, straightforward and non-invasive method to stratify patients and identify appropriate treatments for different disease endotypes.
One of the main distinctions between asthma cases is the type of immune cells involved in causing inflammation. Cases that are primarily eosinophilic typically respond well to glucocorticoid steroid treatments. By contrast, cases dominated by neutrophils show a poor response to steroids. In a previous in vitro study, Schleich et al. identified a selection of volatile organic compounds (VOCs) that could distinguish eosinophilic and neutrophilic asthmas in culture. In this paper, Schleich et al. have attempted to produce comparable results using in vivo breath samples.
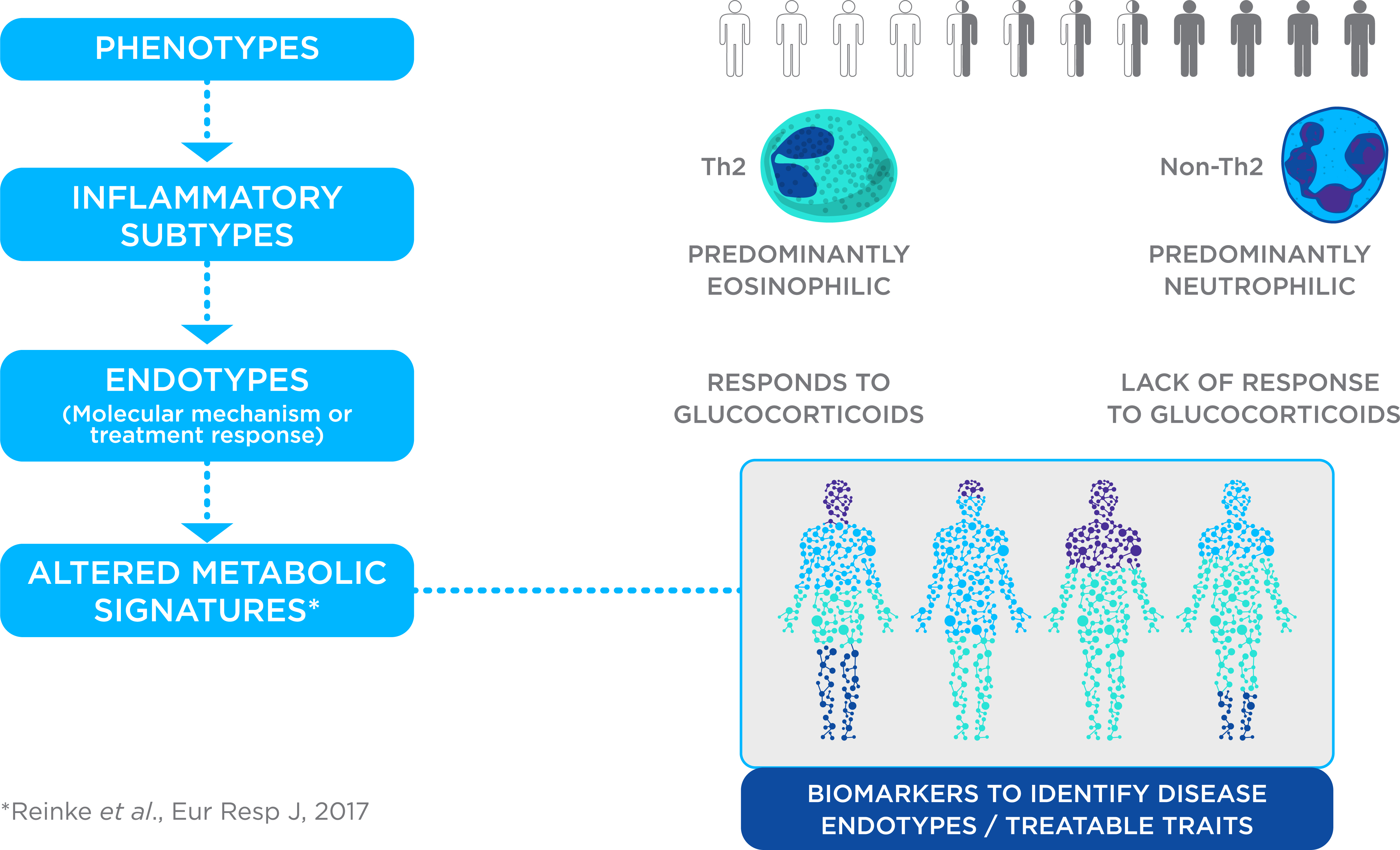
Figure 1. Precision Medicine for Asthma. Asthma management is challenging due to multiple phenotypes, inflammatory subtypes and endotypes with different underlying molecular mechanisms or responses to treatment. Metabolomics is being explored as a route to discover biomarkers to identify disease endotypes or treatable traits, enabling a precision medicine approach to asthma treatment.
Attempting to stratify asthma patients with four VOCs
More than 500 asthma patients were recruited and gave breath samples at one site in Liege, Belgium. Around half of the subjects were used for a discovery study, with the second half included in validation. Participants were associated with asthma inflammatory endotypes on the basis of induced sputum testing – a test that is time-consuming, complex and not suitable for young children, but that is currently the best method for asthma stratification.
The discovery study used GC-MS analysis to identify suitable VOCs and develop predictive algorithms based on those results. The replication study applied GCxGC-HRTOFMS to for a focused analysis of the VOCs that had been selected, applying the algorithms developed as part of the discovery study to a new distinct set of data.
Schleich et al. identified four VOCs as key identifiers of asthma types in the discovery study. Patients with neutrophilic asthmas exhaled elevated levels of nonanal, 1-propanol and hexane relative to other phenotypes, while eosinophilic asthmas could be characterised by lower levels of hexane and 2-hexanone.
In both studies, nonanal proved to be a key predictor for direct discrimination of eosinophil vs. neutrophil phenotypes. The discovery model, which also included 3,7-dimethylnonane and 1-propanol, achieved 70% accuracy, 45% sensitivity and 85% specificity with an area under receiver operating characteristic curve (AUROC) of 0.72.
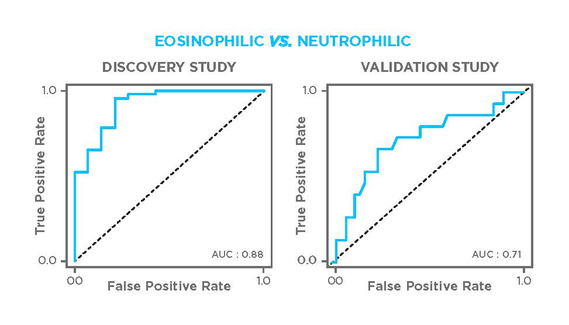
Figure 2. ROC curves representing models for discriminating eosinophilic and neutrophilic asthmas in the discovery (left) and valudation (right) cohorts. The discovery model uses data from VOCs identified as 3,7-dimethylnonane, nonanal and 1-propanol, while the validation model uses hexane and nonanal.
Schleich et al. compared VOC measurement to FeNO and blood eosinophil readings, a diagnostic method suggested by other studies, and found comparable accuracy. Combining all three readings enabled the team to achieve an AUROC of 0.87 (accuracy 76%, sensitivity 79%, specificity 78%) in the replicate cohort.
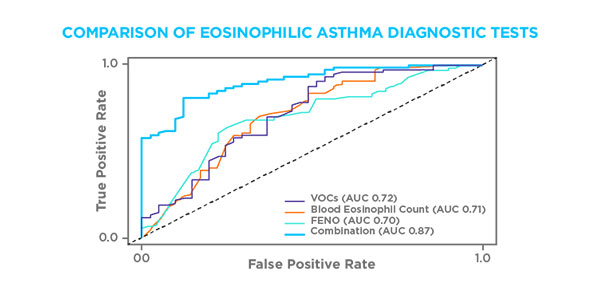
Figure 3. Receiver operating characteristic curve analyses of the sensitivity and specificity of volatile organic compounds (VOCs) (hexane and 2-hexanone), blood eosinophil levels, exhaled nitric oxide fraction (FeNO), and combined VOCs, blood eosinophils, and FeNO for the diagnosis of sputum eosinophilia in the replication cohort. AUC = area under the curve.
Neutrophilic asthma has, in the past, been more difficult to reliably test for. This study distinguished neutrophilic asthma with an AUROC of 0.73 (76% accuracy, 81% sensitivity and 43% specificity), comparable to the statistical power of the aforementioned predictors eosinophilic asthma. Combining the VOC data with preselection on the basis of neutrophil sputum testing they achieved an AUROC of 0.97 (sensitivity 71%, specificity 100%).
Schleich et al. have demonstrated the power of a relatively small number of VOCs to effectively and non-invasively stratify asthma patients. This is an important step towards the use of breath analysis in the diagnosis and treatment of asthma worldwide.
The VOCs used to stratify patients in this study are included in Owlstone Medical’s respiratory disease breath biomarker panel. The Breath Biopsy Panel for Respiratory Diseases is intended to support research to distinguish between types of chronic inflammatory airway diseases and to facilitate better therapeutic decision making and monitoring. The Panel can be integrated into your research as part of the Breath Biopsy OMNI Assay.
References
- The Global Asthma Report, The Global Asthma Network (2018): https://www.globalasthmareport.org/index.html
