What is the Best Method for Breath Biomarker Discovery?
Published on: 16 Mar 2021

Breath researchers have explored a wide range of approaches to identify breath biomarkers and develop breath tests for various clinical applications. As the most versatile and sensitive method, gas chromatography coupled to mass spectrometry (GC-MS) is the gold standard for discovering volatile organic compound (VOC) biomarkers from breath samples.
There is a great interest in developing non-invasive diagnostic tools for disease detection that support the development and use of treatments. Increasingly, studies in this area have focused on VOCs as biomarkers of metabolic and disease processes. Currently, few VOC biomarkers have been reliably identified and validated, and more robust biomarker discovery and validation studies are needed. Accurate VOC detection and identification requires advanced analytical techniques, of which GC-MS is the most established and most widely used. However, a wide range of alternatives have been explored because of their perceived benefits.
Other Methods for Breath Biomarker Discovery
Other methods are often favored because they are easier than GC-MS to use in a clinical setting. They largely fall into two groups: simpler variants of mass spectrometry, and sensor-based systems. Popular options in the first group include selected ion flow tube mass spectrometry (SIFT-MS), proton transfer reaction mass spectrometry (PTR-MS), and the more distantly related ion mobility spectrometry (IMS). Sensors for VOC detection have also become known as electronic noses (eNoses) and operate in a similar way to a biological nose, with different sensors responding to different chemical groups.
More About the Different Methods of Breath Analysis
Sensitivity, reproducibility and consistency of results are all key priorities to consider when selecting a method for biomarker discovery. Limitations such as cost, time and available skills are also important. A further factor is clinical relevance, but this can be misleading at the biomarker discovery stage, where reliable results are more important than ease of use.
Read our blog of tips for study design
All analysis methods have their own strengths and limitations, and all have various parameters that can be altered for specific needs. While GC-MS is the established gold standard for breath analysis, no single method can be relied upon to provide a definitive and complete list of all the VOCs found in a breath sample.
Advantages of GC-MS for Breath Biomarker Discovery
Reproducible biomarker discovery
Some techniques, particularly those based on sensors, use holistic ‘breathprints’ to differentiate groups of samples. While this has been effective in individual studies, these approaches can struggle to provide reproducible results due to the lack of insight into the biomarkers that make up each breathprint. As such, breathprints from different studies can vary and there’s no way to investigate the underlying cause.
With the help of compound libraries, GC-MS can identify, and even quantify, individual VOC biomarkers, which offers a range of benefits. Known biomarkers are easier to compare across studies and they can be related to underlying disease processes. The extra evidence this provides can help biomarkers to gain acceptance and is a valuable foundation for clinical implementation of breath tests. Analytical devices are rarely perfectly stable – knowing biomarker identities also has technical benefits in helping to calibrate systems and normalize data, as well as providing insights that can be used to optimize biomarker detection.
Breadth of biomarker detection
Breath is a complex sample source containing as many as 1000 different VOCs with abundances that vary by as much as six orders of magnitude (106). GC-MS is uniquely well equipped to analyze such challenging samples. The latest HRAM GC-MS devices (such as GC-Orbitrap™) have the resolution to reliably identify individual compounds within complex mixtures. They also offer dynamic ranges that span the range of VOCs on breath alongside uniquely sensitive detection, in the parts per trillion (ppt) range, across the breadth of detectable compounds.
An optimized HRAM GC-MS system can detect hundreds of compounds in a breath sample and demonstrate high consistency in detecting the same compounds across samples. They can also be further adapted and iteratively optimized, based on existing results and biological insights, to enhance the detection of particular compounds. For example, the gas chromatography column can be changed to separate compounds using different chemical properties.
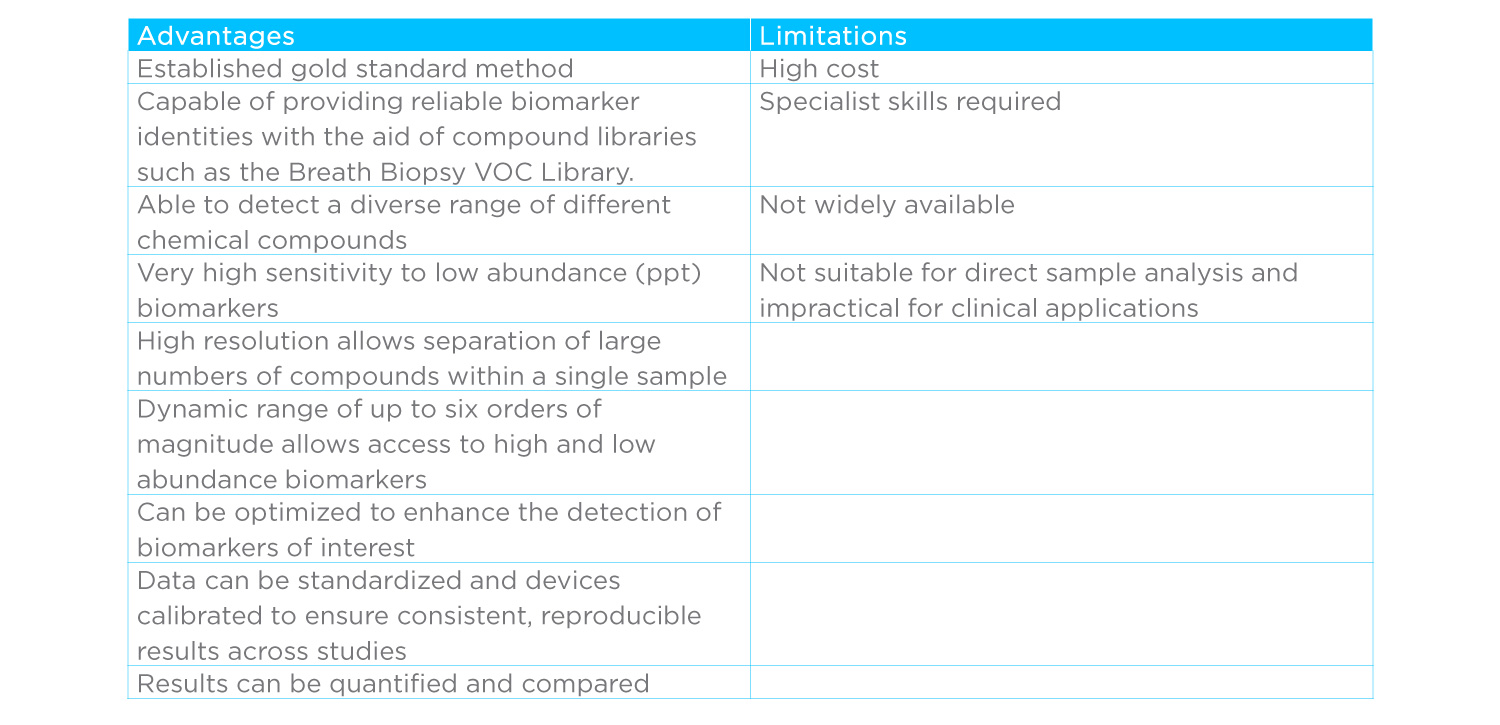
Limitations of GC-MS for Biomarker Discovery
GC-MS devices are expensive, slow, complex and require specialist skills to use and to interpret results. As such they’re also not widely available. Notably, SIFT-MS and PTR-MS devices are also costly but are more suited to use in the clinic due to their relative ease of use. The desire is for affordable, easy to use clinical tests that provide clear and immediate (or at least rapid) results, is one reason why studies have sought alternatives to GC-MS.
Yet, any breath test with a chance of becoming widely used in the clinical setting will undoubtedly need to be firmly underpinned by well characterized biomarkers supported by a robust body of research and biological evidence. It will need to provide consistent and reproducible results independent of context. As such, while GC-MS isn’t suited to online testing that provides an immediate result, it has a vital part to play in discovering and validating biomarkers as a critical aspect of establishing their clinical relevance. What’s more, in most settings, it is more important for results to be accurate than fast, in these cases a test that sends samples to a specialist centralized facility for analysis is more useful and offers economies of scale that make the use of GC-MS analysis financially preferable.
The strength of HRAM GC-MS as a tool for biomarker discovery is why we use it as the basis for Breath Biopsy®. We have developed the Breath Biopsy OMNI Assay as an optimized method for breath analysis, supported by specialist expertise in GC-MS use and data analysis to provide everyone with access to this gold standard capability. We then also have the expertise to support development of breath tests that meet specific clinical needs, including the possibility of direct point of care applications.
Find out more about Breath Biopsy by downloading the Breath Biopsy: The Complete Guide for more information about the applications of Breath Biopsy.