Owlstone Medical to provide Breath Biomarker Services for 4D Pharma using Breath Biopsy® platform
Partnership aims to identify non-invasive VOC biomarkers in development of live biotherapeutics for disease diagnosis and patient stratification
Published on: 7 Mar 2017
Partnership aims to identify non-invasive VOC biomarkers in development of live biotherapeutics for disease diagnosis and patient stratification
Cambridge, UK, March 7, 2017: Owlstone Medical, a diagnostics company developing a breathalyzer for disease, today announced it has signed a master service agreement with 4D Pharma plc (AIM:DDDD), a pharmaceutical company focusing on the development of live biotherapeutics. As part of the agreement, 4D Pharma will use Owlstone Medical’s Biomarker Services to discover novel non-invasive biomarkers present in breath that can be applied to the development of effective therapeutics for a wide range of diseases from cancer, to gastrointestinal, autoimmune, and respiratory conditions.
The pattern of Volatile Organic Compounds (VOCs) present in exhaled breath is directly linked to the metabolic processes taking place within the body as well as exogenous sources such as an individual’s microbiome, the many millions of bacteria that colonize the body and play an important role in health and disease.
4D Pharma is developing live biotherapeutics, a revolutionary class of medicines that harness the functionality of the microbiome. The company is using Owlstone Medical’s Biomarker Services to identify novel VOC biomarkers in breath for use in disease diagnosis and stratification to match patients to the most appropriate treatment.
Breath Biopsy® samples will be collected from patients at 4D Pharma’s clinical trial site using Owlstone Medical’s ReCIVA Breath Sampler. Breath samples stored in stable sorbent tubes are then transferred to Owlstone Medical’s service laboratory in Cambridge, UK, for VOC analysis using the company’s FAIMS platform to analyze spectral data and develop classification algorithms.
Billy Boyle, co-founder and CEO at Owlstone Medical, said: “Our Biomarker Services draw on the experience we have developed through our pioneering work on Breath Biopsy technology and the large breath biomarker clinical trials we are running as part of our own clinical pipeline. We are excited to enable 4D Pharma to discover VOC biomarkers for patient stratification in their own therapeutic programs.”
Professor Paul O’Toole at 4D Pharma Cork Ltd commented: “We are very pleased to initiate this Breath Biomarker Services project with Owlstone Medical. VOCs in breath provide incredibly valuable information about the health of an individual as well as potentially reflecting the metabolism of their microbiome. As such, VOC biomarkers show great potential for patient stratification and could help us to determine which patients would benefit most from our live biotherapeutics.”
To find out more about Owlstone Medical visit: www.owlstonemedical.com
ENDS
Notes to Editors
 |
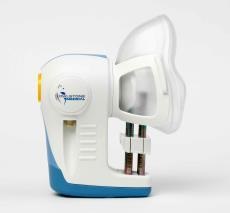 |
Photo: Breath collection – sorbent storage tubes and ReCIVA Breath sampler
For a high resolution images please contact sarah.jeffery@zymecommunications.com
Media contact:
For more information please contact:
Sarah Jeffery, Zyme Communications
sarah.jeffery@zymecommunications.com
+44 (0)7771 730 919
About Owlstone Medical
Owlstone Medical has developed a breathalyzer for disease. With a focus on non-invasive diagnostics for cancer, inflammatory disease and infectious disease, the company aims to save 100,000 lives and $1.5B in health care costs by 2020.
The company’s microchip FAIMS (Field Asymmetric Ion Mobility Spectrometer) sensor is a platform technology that can be programmed in software to detect targeted biomarkers of disease in breath (and other bodily fluids). FAIMS can be used in clinical diagnostics and precision medicine with application in cancer and a wide range of other medical conditions. Highly sensitive and selective, these tests allow for early diagnosis when treatments are more effective and where more lives can be saved.
Owlstone Medical is currently developing tests for lung and colorectal cancer, two of the most common cancer killers worldwide, and for asthma stratification by therapeutic response. The company also sells R&D tools and services to academic, clinical and pharma partners who want to develop breath based diagnostics for their own applications.
Founded in 2004 as a spin-out from the Engineering Department at the University of Cambridge, Owlstone Inc is a profitable business selling its proven FAIMS technology to military and industrial customers globally. Owlstone Medical was spun out from Owlstone Inc in 2016 to develop and commercialise FAIMS in diagnostic applications.
Owlstone Medical is headquartered in Cambridge, UK, with offices in London, UK.
