ReCIVA® Breath Sampler
Reliable and reproducible, non-invasive breath sample collection
- Collect volatile organic compound and respiratory droplet samples from exhaled breath
- Simple, non-invasive sampling during normal tidal breathing with options for breath fraction targeting
- Designed and optimized to ensure high patient safety and comfort
- Used in over 100 academic and clinical research sites worldwide
INTRODUCTION
Developed by experts in breath research to optimize breath sample collection
Breath is a rich and complex sampling medium containing both VOCs and respiratory droplets carrying non-volatiles, proteins, lipids, viruses, and even bacteria. As with any analytical challenge, accessing the information contained in breath depends on highly reproducible analysis supported by standardized sample collection. These are vital to maximizing the comparability and consistency of results, which is essential to driving progress and achieving widespread clinical implementation of breath research.
ReCIVA was developed in collaboration with multi-disciplinary leaders in the breath research field as an optimized tool for robust and reliable breath sample collection that maximizes clinical applications by prioritizing safety and comfort during use.
Breath Biopsy Products and Services are for research use only. Not for use in diagnostic procedures.
- ROBUST AND REPRODUCIBLE
Each Breath Biopsy Cartridge includes four independent adsorbent tubes for simultaneous VOC collection, providing multiple experimental replicates
- COMFORTABLE AND SAFE
ReCIVA is non-invasive, safe and easy to use, making it suitable for breath sampling with almost any subject group across a wide range of disease contexts
- SELECTIVE
Breath pattern recognition means ReCIVA can collect specific breath fractions, allowing targeted analysis from different regions of the airways
- SENSITIVE
Pre-concentration of VOCs directly from breath onto adsorbent tubes allows more VOCs to be captured per sample increasing the sensitivity of the system to low abundance VOCs
- ANALYSIS OPTIONS
VOC samples can be analyzed with a range of tools including GC-MS and FAIMS while respiratory droplets are compatible with established analytical processes
- FLEXIBLE
The quality of seal produced by the standard ReCIVA mouthpiece provides a high degree of flexibility for use with various face shapes.
HOW IT WORKS
Simultaneous sample collection and pre-concentration
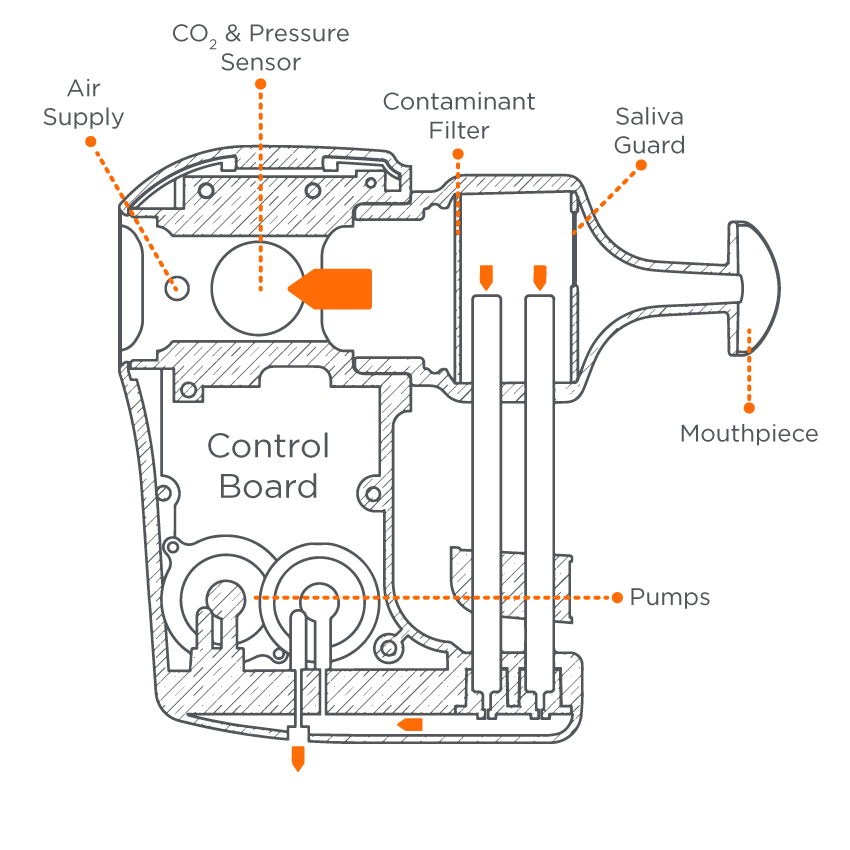
Whatever your area of research interest, we’ve designed ReCIVA to be as versatile as possible while still providing quality breath sample collection. ReCIVA simultaneously collects replicate samples contributing to the reliability of Breath Biopsy and the robustness of results. Each Breath Biopsy Cartridge includes four independent adsorbent tubes for VOC collection. Tubes can be combined to enhance sensitivity or analyzed separately as replicate samples.
ReCIVA directly captures and pre-concentrates VOC biomarkers in breath onto the Breath Biopsy Cartridge. The direct extraction of VOCs from atmospheric gases means samples can be greatly enriched for VOCs on breath, even those at low abundance, simply by collecting samples for longer. This pre-concentration means that Breath Biopsy can offer higher sensitivity for trace VOCs, it also means Breath Biopsy samples are more robust during shipping or storage before sample analysis.
SELECTIVE COLLECTION
Real time feedback and breath fraction collection
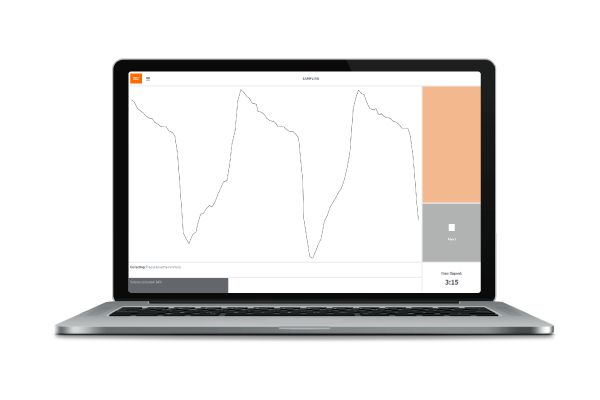
By adjusting ReCIVA’s collection settings using the straightforward Breath Biopsy Collect software interface, you can also collect VOCs from selected breath fractions allowing you to differentially analyze VOCs originating from the deep lungs, upper airways or mouth and throat, depending on your area of interest.
ReCIVA’s built-in carbon dioxide and pressure sensors monitor and learn the breath patterns of each subject in real time and only activate the pumps that draw air into the Breath Biopsy Cartridge at the right times to collect each breath fraction (e.g. end tidal or bronchial airways).
BREATH ANALYSIS
Analysis of VOCs on breath

ReCIVA is used to collect breath samples onto Breath Biopsy Cartridges for separate analysis. It is ideal for use in conjunction with techniques such as GC-MS (gas chromatography-mass spectrometry) and GC-FAIMS (gas chromatography field asymmetric ion mobility spectrometry).
Breath Biopsy OMNI enables you to collect samples with ReCIVA and send them to us for global biomarker discovery analysis, as part of a complete package that gives you access to our expert team for study planning, operation, data analysis and interpretation.
Opting to use OMNI includes VOC detection using high resolution accurate mass (HRAM) mass spectrometry and high confidence VOC identity assignment using our HRAM Breath Biopsy Library and the NIST library. Our Library includes many compounds on breath with evidence of associations to various key diseases and biological processes.
Breath Biopsy offers complete support for sample collection and analysis. Find out about our analysis services