Detection of colorectal cancer by analysis of urinary VOCs
FAIMS could prove valuable in the future as part of a screening program for early-stage bowel cancer
| Publication information: Arasaradnam et al., Detection of Colorectal Cancer (CRC) by Urinary Volatile Organic Compound Analysis, PLoS ONE. 9(9): e108750. DOI: 10.1371/journal.pone.0108750
Disease Area: Colorectal cancer Application: Early detection Sample medium: Urine Products: Lonestar® VOC Analyzer, ATLAS Headspace Sampler Analysis approach: FAIMS, GC-MS Summary:
|
Bowel cancer (also known as colorectal cancer) is the fourth most common form of cancer in the UK, with 75 new cases per 100,000 men and 57 new cases per 100,000 women each year. It is the second most common cause of death from cancer in the UK, and has a mortality rate of around 45% within five years of diagnosis. As with many other types of cancer, the stage at which bowel cancer is diagnosed has a huge impact on the patient’s prognosis. Patients identified at Stage I have around a 97% five-year relative survival rate, while those identified at Stage IV have around a 7.5% five-year relative survival rate.
To increase the number of early diagnoses, the way in which patients are screened for colorectal cancer needs to be improved. Existing non-invasive colorectal cancer screening methods such as gFOB (guaiac based fecal occult blood) and the now more common FIT (Fecal Immunochemical Testing) show relatively low sensitivities (FIT sensitivity = 66 – 88%, specificity = 87 – 96%). FIT also lacks sensitivity to advanced adenoma (27 – 41%, specificity = 91 – 97%). A major drawback of these techniques is that patients have to provide stool samples. Arasaradnam et al. note that in their locality, the uptake of screening utilizing fecal samples is only ~50%.
In this study it was found that FAIMS was used to differentiate between patients with colorectal cancer and healthy controls, with a sensitivity of 88%, by rapid, non-invasive analysis of volatile organic compounds (VOCs) in urine. Detection of colorectal cancer’s ‘bio-odorant fingerprint’ with FAIMS from patient urine samples represents a major step forward in simple, non-invasive diagnosis.
The authors recruited 133 volunteers, 83 of whom had confirmed colorectal cancer. Urine samples were taken, and the volatile organic compounds (VOCs) present were analyzed using FAIMS and gas chromatography mass spectrometry (GC-MS). FAIMS analysis was performed using an Owlstone Medical Lonestar® VOC analyzer fitted with an ATLAS sampling system.
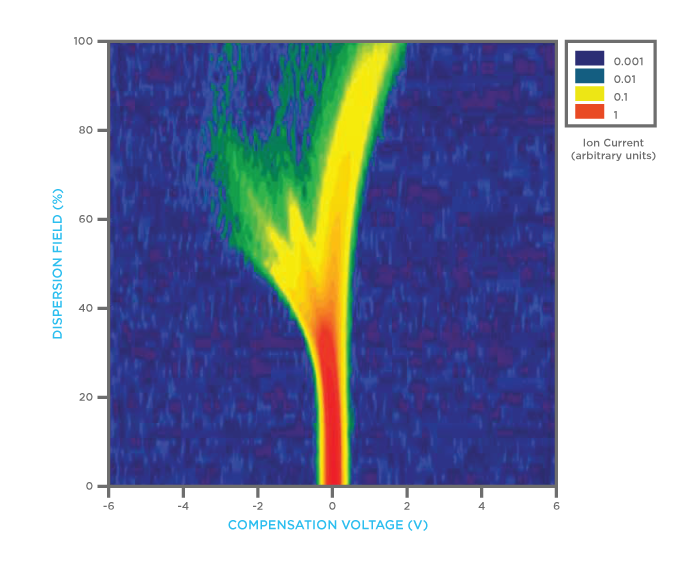
FAIMS spectrum from urine sample of patient with colorectal cancer
FAIMS is a physical measurement of molecules rather than a measurement of chemical interaction, which is the case for instruments that use traditional ‘eNose’ chemical sensor technology. For this reason, FAIMS spectra are very information rich (see figure above, which shows a typical FAIMS spectra from a urine sample taken from a patient with confirmed colorectal cancer). Multivariate data analysis techniques allow the simple interpretation of complex FAIMS data to determine if differences in groups can be detected.
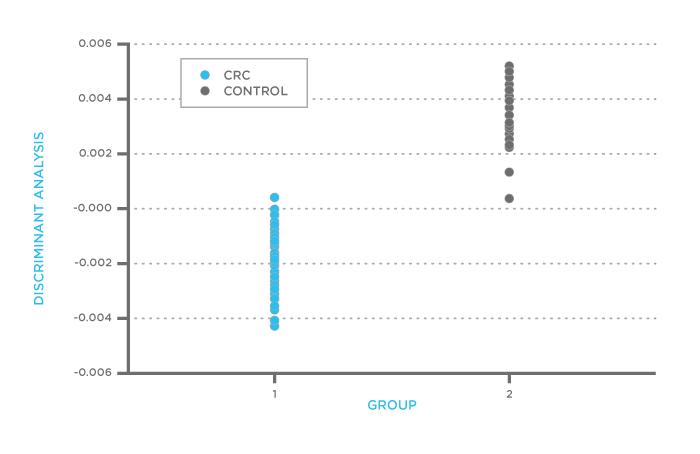
Fisher Discriminant Analysis (FDA) of FAIMS data from patients with colorectal cancer and controls
To determine whether the patients with colorectal cancer could be distinguished from the other healthy volunteers using FAIMS, Arasaradnam et al. used Fisher Discriminant Analysis on the data collected from the patient urine samples. They found that it was possible to use the entire VOC profile measured with FAIMS to detect colorectal cancer cases with 88% sensitivity. These initial results show that FAIMS can prove valuable as part of a screening program for early-stage bowel cancer.


