Breath Biomarker discovery for C. difficile
VOCs produced metabolically could be useful to diagnose this infection
| Publication information: Teny John, Leen Hasan, Kirk Pappan, Owen Birch, David Grove, Gary Procop, Raed Dweik, Billy Boyle, Max Allsworth, Nabin Shrestha ‘Volatile Compound Analysis of Breath from Patients with Clostridium difficile Infection for Biomarker Discovery and Biological Insight – View Poster
Disease Area: C. difficile Application: Diagnostics Sample medium: Breath Products: ReCIVA® Breath Sampler, Breath Biopsy® Services Analysis approach: TD-GC-MS Summary:
|
C. difficile is one of the leading causes of hospital-acquired diarrhea, representing a significant issue for hospital management. Current or recent antibiotic treatment is the main risk factor of contracting the infection. While diarrhea is the main symptom, certain co-morbidities can make C. difficile infections (CDIs) potentially life-threatening.
Evidence that specific volatile organic compounds (VOCs) may be produced in the human metabolome as a consequence of CDI suggests that breath could be a useful diagnostic tool. Owlstone Medical worked with Cleveland Clinic to try and identify a selection of VOCs on exhaled breath capable of discriminating between patients with and without CDI and to obtain biological insight into CDI associations and risk factors.
Methods
Breath samples were collected from each patient at the Cleveland Clinic, using the ReCIVA Breath Sampler, after diagnosis from a population of hospitalized patients with diarrhea. Subjects with diarrhea who tested positive for a CDI via PCR test were added to the CDI group and their samples were matched demographically as closely as possible against control subjects who also had diarrhea but had tested negative for CDI. There were 17 patients in each group.
Samples were then analyzed by Owlstone Medical in the Breath Biopsy Laboratory. Thermal desorption-gas chromatography-mass spectrometry (TD-GC-MS) chromatograms were converted into molecular feature (MF) lists for statistical analysis.
Results
Sixty-five volatile compounds were included in the final curated data set and 14 were assigned a tentative identification based on matching to the National Institute of Standards and Technology (NIST) database (Table 1).
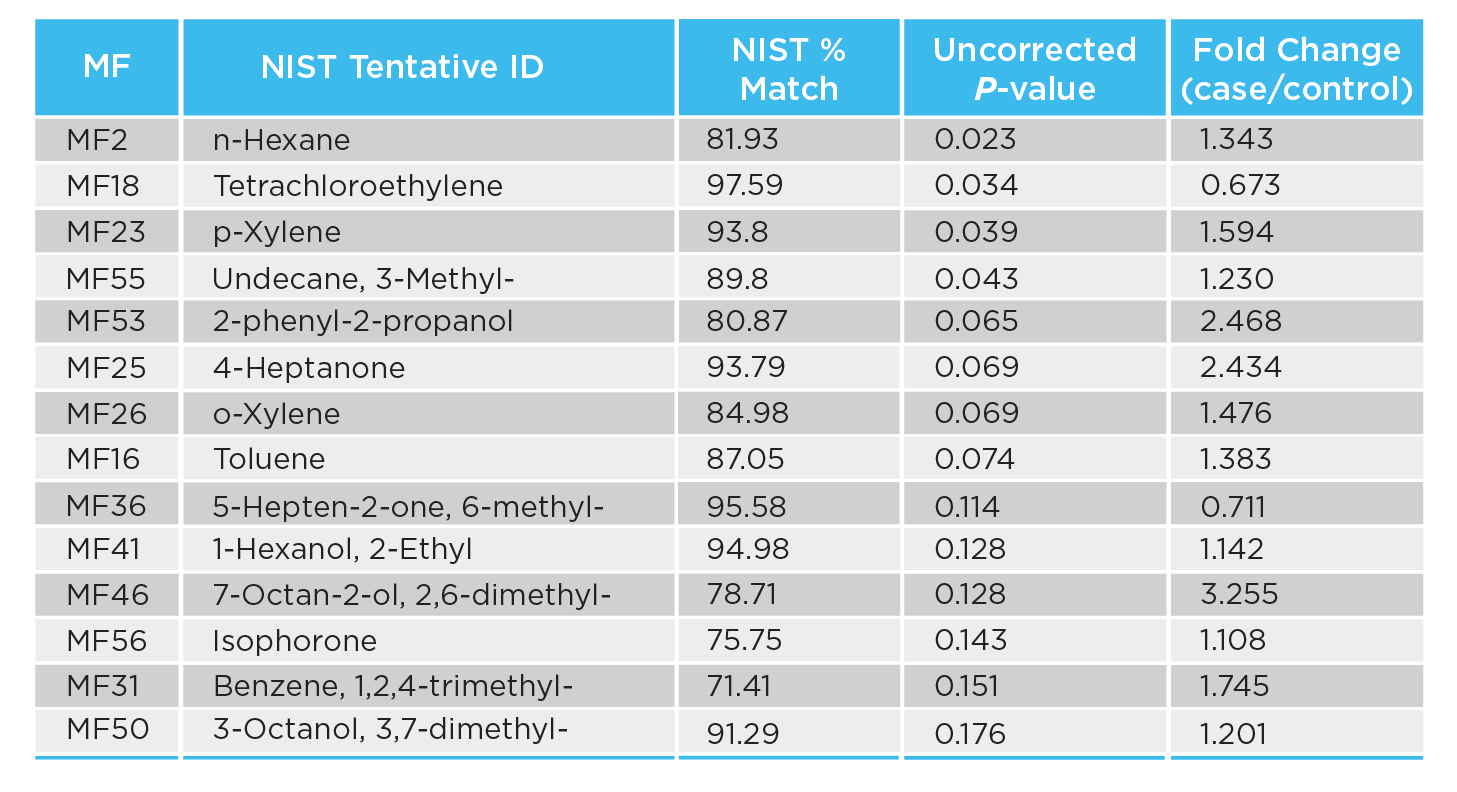
Table 1: Fourteen volatile compounds with tentative identifications are shown along with their NIST scores and Mann-Whitney U-test P values.
Nine tentatively identified VOCs were selected by a quadratic discriminant analysis modeling method. The model was able to distinguish between CDI cases and controls with 0.74 accuracy, sensitivity of 0.71, a specificity of 0.76, and a mean area under the receiver operating characteristic curve (AUROC) of 0.72 (Fig. 1).
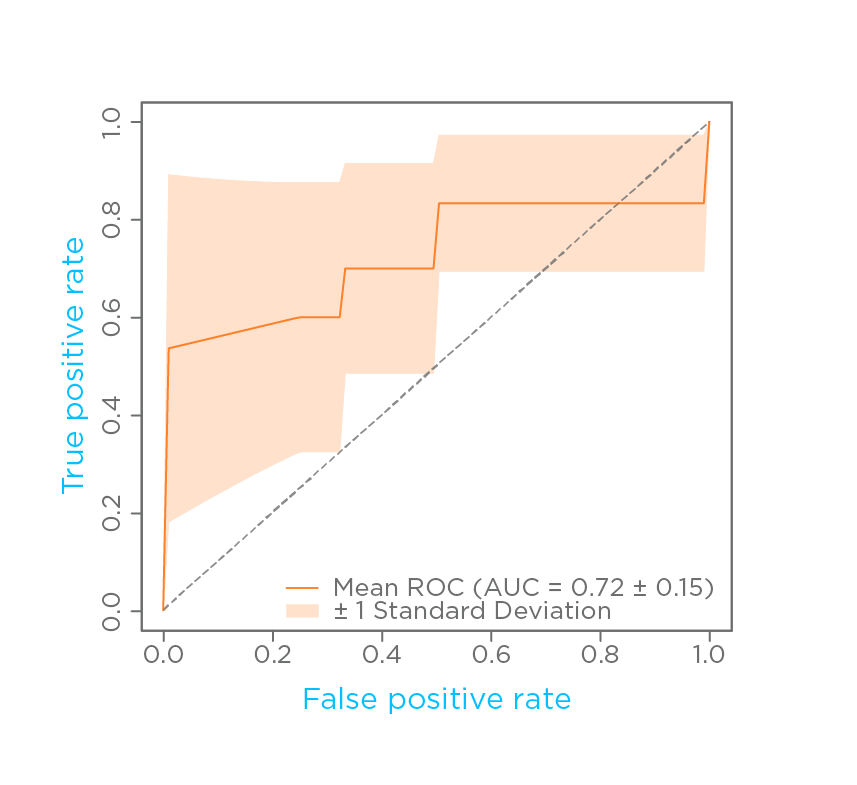
Figure 1: Receiver operating characteristic (ROC) curve for the model with 9 VOCs had an accuracy 0.74, AUC 0.72, sensitivity 0.71, and specificity 0.76.
Lipid peroxidation, as potentially indicated by statistically significant increases of the 2 alkanes n-Hexane and 3-methylundecane, appeared to be higher in CDI patients than in controls.
Some Clostridium species have been previously reported to metabolize tetrachloroethylene, which tallies with the relatively significant decrease of tetrachloroethylene in CDI patients found in this study. This could be further investigated and confirmed in vitro.

